Point-Of-Care Molecular Diagnostics Market Size, Share, Trends, Growth 2034

Point-Of-Care Molecular Diagnostics Market By Product Type (Instruments and Analyzers, Reagents and Consumables, Software and Services), By Technology (Polymerase Chain Reaction, Isothermal Amplification, Hybridization, Next-Generation Sequencing), By Application (Infectious Disease Testing, Oncology and Cancer Screening, Cardiology, Neurological Disorders, Sexual Health and Sexually Transmitted Infections), By End-User (Hospitals and Clinics, Diagnostic Laboratories, Ambulatory Care Centers, Home Care Settings, Pharmacies), By Distribution Channel (Direct Sales, Distributors and Dealers, Online Retail), and By Region – Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, and Forecasts 2025 - 2034-
| Market Size in 2024 | Market Forecast in 2034 | CAGR (in %) | Base Year |
|---|---|---|---|
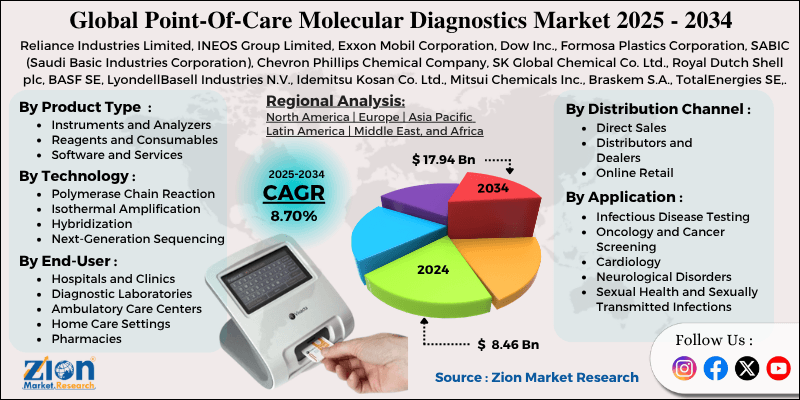
| USD 8.46 Billion | USD 17.94 Billion | 8.70% | 2024 |
Point-Of-Care Molecular Diagnostics Industry Perspective:
What will be the size of the point-of-care molecular diagnostics market during the forecast period?
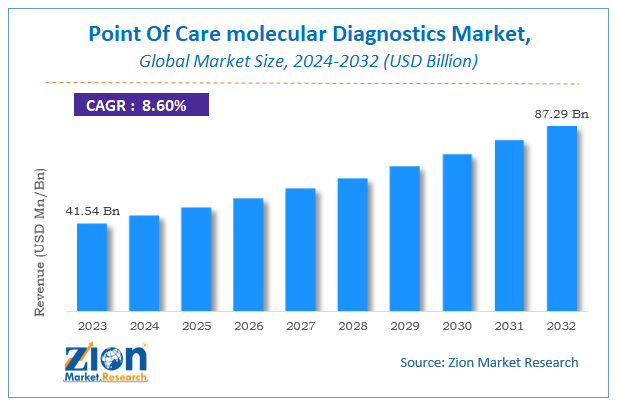
The global point-of-care molecular diagnostics market size was worth approximately USD 8.46 billion in 2024 and is projected to grow to around USD 17.94 billion by 2034, with a compound annual growth rate (CAGR) of roughly 8.70% between 2025 and 2034.
 Request Free SampleRequest Free SampleRequest Free Sample
Request Free SampleRequest Free SampleRequest Free Sample
Key Insights
- As per the analysis shared by our research analyst, the global point-of-care molecular diagnostics market is estimated to grow annually at a CAGR of around 8.70% over the forecast period (2025–2034).
- In terms of revenue, the global point-of-care molecular diagnostics market size was valued at approximately USD 8.46 billion in 2024 and is projected to reach USD 17.94 billion by 2034.
- The point-of-care molecular diagnostics market is poised for strong growth, driven by rising infectious disease rates, demand for rapid diagnostics, increased healthcare investment, advancing affordable technologies, and expanding use across multiple disease areas.
- Based on product type, the reagents and consumables segment is expected to lead the point-of-care molecular diagnostics market, while the instruments and analyzers segment is anticipated to grow rapidly.
- Based on technology, the polymerase chain reaction segment is expected to lead the point-of-care molecular diagnostics market, while the isothermal amplification segment is anticipated to grow at the fastest rate.
- Based on application, the infectious disease testing segment is expected to lead the point-of-care molecular diagnostics market, while the oncology and cancer screening segment is anticipated to grow notably.
- Based on end-user, the hospitals and clinics segment is the largest current end-user group, while the home care settings segment is anticipated to grow most rapidly.
- Based on distribution channel, the direct sales segment is expected to lead the point-of-care molecular diagnostics market, while the online retail segment is anticipated to grow steadily.
- Based on region, North America is projected to dominate the global point-of-care molecular diagnostics market during the estimated period, followed by Europe.
Point-Of-Care Molecular Diagnostics Market: Overview
Point-of-care molecular diagnostics is a type of medical testing that allows doctors to detect diseases at or near the patient’s location using genetic material, such as DNA or RNA. Instead of sending samples to a distant laboratory and waiting for results, these tests are performed on-site, providing quick results. They can accurately identify viruses, bacteria, and other infections. Most tests give results within minutes to a few hours, making them much faster than traditional laboratory tests, which may take a day or longer. The main idea behind point-of-care testing is to help doctors make quick and accurate decisions. When results are available immediately, doctors can start the right treatment without delay. This reduces the risk of incorrect diagnosis and prevents unnecessary antibiotic use. It also helps control the spread of infectious diseases, as patients can be treated or isolated right away.
Over the years, point-of-care molecular diagnostics technology has advanced significantly. Devices have become smaller, faster, and easier to use, even for non-specialists. They are now used in many places such as hospitals, clinics, pharmacies, rural health centers, and sometimes even at home.
The rising burden of infectious diseases, growing demand for rapid diagnostics, increased investment in decentralized healthcare, and advances in miniaturized testing are driving strong growth in the point-of-care molecular diagnostics market.
Point-Of-Care Molecular Diagnostics Market: Technology Roadmap 2025–2034
What is the projected development roadmap of the point-of-care molecular diagnostics market over the forecast period?
The point-of-care molecular diagnostics market is evolving with advances in miniaturized platforms, isothermal chemistry, AI integration, multiplex testing, and expansion into community and home settings. The market is expected to grow at a CAGR of around 8.70% over the forecast period, driven by strong demand across infectious disease management, cancer screening, sexual health, and personalized medicine segments.
The following roadmap outlines key development phases expected through 2034.
2025–2027: Miniaturization and Faster Testing Phase
- Diagnostic devices are expected to become smaller and portable, enabling testing in remote settings without large laboratory equipment.
- Testing time is likely to drop further, with many devices delivering results in under fifteen minutes for common infections like flu and throat infections.
- Devices are projected to include built-in connectivity, enabling automatic sharing of results with doctors and health systems in real time.
2028–2031: Multiplex Testing and Wider Use Phase
- Testing platforms are expected to detect multiple diseases from a single sample, reducing the need for repeat tests and speeding up treatment.
- These diagnostics are likely to expand beyond infections into areas such as early cancer detection, heart disease risk assessment, and personalized medicine.
- Artificial intelligence is projected to support doctors by helping interpret results and suggesting next steps.
2032–2034: Integrated and Home-Based Testing Phase
- Diagnostic devices are expected to connect fully with digital health systems, linking results to hospitals and telemedicine platforms.
- Advanced testing methods are likely to move closer to patients, enabling detailed disease analysis and treatment decisions at the point of care.
- Home testing kits with smartphone connectivity are projected to become common, allowing people to test themselves and share results directly with doctors.
Point-Of-Care Molecular Diagnostics Market: Dynamics
Growth Drivers
How is the rising global burden of infectious diseases and the demand for rapid diagnosis driving growth in the point-of-care molecular diagnostics market?
The point-of-care molecular diagnostics market is growing steadily due to the rising burden of infectious diseases and the need for faster diagnosis across healthcare settings. Diseases such as respiratory infections, tuberculosis, sexually transmitted infections, and emerging viral outbreaks require rapid and accurate testing to control their spread and start treatment promptly. Traditional laboratory testing often takes longer, delaying care and increasing healthcare costs. Point-of-care molecular diagnostics solve this by providing fast, highly accurate results directly in clinics, hospitals, and even remote health centers, improving patient management and reducing repeat visits. This ability to deliver lab-quality results quickly is a major factor driving adoption across both developed and developing regions. The COVID-19 pandemic further highlighted the importance of rapid testing, leading to increased investment in decentralized healthcare systems and diagnostic infrastructure. As a result, demand for faster, reliable, and accessible testing solutions continues to rise, strongly supporting the growth of the point-of-care molecular diagnostics industry worldwide.
The shift toward decentralized, community-level healthcare is driving the point-of-care molecular diagnostics market.
The point-of-care molecular diagnostics market is growing due to a major shift toward decentralized healthcare, in which services are delivered closer to patients rather than in large centralized laboratories. In the past, people often had to travel far or wait days for test results, which delayed treatment and increased costs. Today, governments and healthcare providers are investing in community clinics, mobile health units, pharmacy-based testing, and telehealth services that require fast and reliable diagnostics. Point-of-care molecular diagnostics support this shift by providing accurate results at the site of care without the need for complex laboratory infrastructure. This improves access, speeds up decision-making, and enhances patient outcomes. The demand is even stronger in developing regions like South Asia, Southeast Asia, and Africa, where laboratory facilities are limited in rural areas. Portable, battery-powered testing devices are enabling advanced diagnostics in remote locations. This expanding access, combined with rising healthcare investments and the need for faster services, is a key driver supporting the growth of the point-of-care molecular diagnostics market worldwide.
Restraints
The high costs, reimbursement issues, and skill requirements limit adoption in the point-of-care molecular diagnostics market.
The point-of-care molecular diagnostics market faces several challenges that slow its adoption, particularly in smaller, resource-limited healthcare settings. One major issue is the high cost of diagnostic devices, which require a large upfront investment that many small clinics and community health centers cannot easily afford. In addition, the ongoing cost of test materials and consumables adds financial pressure and makes regular use more difficult.
Another key challenge is limited reimbursement support, as insurance systems in many regions do not fully cover these tests, making them expensive for both providers and patients. This reduces their usage even when they are clinically beneficial. Training is also an important concern, as healthcare workers need proper skills to handle samples, operate devices, and interpret results correctly. In busy healthcare environments, maintaining trained staff can be difficult due to frequent turnover. These cost, reimbursement, and training challenges act as major restraints, limiting the widespread adoption and growth of the point-of-care molecular diagnostics market.
Opportunities
What opportunities are created by expanding use in cancer and chronic disease in the point-of-care molecular diagnostics market?
The point-of-care molecular diagnostics industry is creating new growth opportunities as its use expands beyond infectious disease testing into broader areas of healthcare. One major opportunity is cancer screening, where these tests can detect early signs of cancer at the genetic level before symptoms appear, improving survival rates and treatment outcomes. Another important area is chronic disease management, including conditions such as diabetes, heart disease, and autoimmune disorders, in which molecular testing helps doctors select appropriate treatments and monitor patients' health more effectively. The rise of personalized medicine is also increasing demand for pharmacogenomic testing, which uses a patient’s genetic information to guide drug selection and dosage. Bringing these tests closer to patients improves speed and convenience in care delivery. In addition, the growing problem of antimicrobial resistance is driving demand for rapid testing that can identify infections and quickly suggest the most effective treatment. These expanding applications and rising clinical needs are creating significant growth opportunities in the point-of-care molecular diagnostics market.
Challenges
Why do regulatory challenges, quality issues, and error risks affect the point-of-care molecular diagnostics market?
The point-of-care molecular diagnostics market faces significant challenges, including stringent regulations and the need to maintain test quality in real-world settings. These devices must pass detailed approval processes before use, and rules differ across countries, making market entry costly and time-consuming for companies. In addition, maintaining high test accuracy outside laboratories is difficult, as tests are often performed by healthcare workers with varying levels of training and in less controlled environments. This increases the risk of errors during testing and result interpretation. Another major concern is the risk of false results, which can lead to incorrect treatments or missed diagnoses. Ensuring consistent performance across different locations and conditions remains a key challenge. These regulatory complexities, quality-control issues, and the risk of inaccurate results pose major barriers, slowing the adoption and expansion of the point-of-care molecular diagnostics market globally.
 Request Free SampleRequest Free SampleRequest Free Sample
Request Free SampleRequest Free SampleRequest Free Sample
Point-Of-Care Molecular Diagnostics Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Point-Of-Care Molecular Diagnostics Market |
| Market Size in 2024 | USD 8.46 Billion |
| Market Forecast in 2034 | USD 17.94 Bllion |
| Growth Rate | CAGR of 8.70% |
| Number of Pages | 225 |
| Key Companies Covered | Reliance Industries Limited, INEOS Group Limited, Exxon Mobil Corporation, Dow Inc., Formosa Plastics Corporation, SABIC (Saudi Basic Industries Corporation), Chevron Phillips Chemical Company, SK Global Chemical Co. Ltd., Royal Dutch Shell plc, BASF SE, LyondellBasell Industries N.V., Idemitsu Kosan Co. Ltd., Mitsui Chemicals Inc., Braskem S.A., TotalEnergies SE, and others. |
| Segments Covered | By Product Type, By Technology, By Application, By End-User, By Distribution Channel, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2034 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Point-Of-Care Molecular Diagnostics Market: Segmentation
The global point-of-care molecular diagnostics market is segmented by product type, technology, application, end-user, distribution channel, and region.
What makes the reagents and consumables segment the expected leader in the point-of-care molecular diagnostics market?
Based on product type, the global point-of-care molecular diagnostics market is categorized into instruments and analyzers, reagents and consumables, and software and services. The reagents and consumables segment holds approximately 58% of the global market share and is expected to remain dominant throughout the forecast period. This segment leads because every single test performed requires a fresh set of reagents and consumables, creating a steady, recurring, and highly predictable revenue stream that grows in direct proportion to testing volume. The instruments and analyzers segment holds around 29% of market share and is growing rapidly, as new product launches and falling hardware prices bring molecular testing within reach of a wider range of healthcare providers.
How does the polymerase chain reaction technology segment maintain its leading position in the point-of-care molecular diagnostics market?
Based on technology, the point-of-care molecular diagnostics market is divided into polymerase chain reaction, isothermal amplification, hybridization, and next-generation sequencing. The polymerase chain reaction segment accounts for approximately 52% of the global market share, driven by its well-established clinical validation, very high sensitivity and specificity, and widespread familiarity among healthcare professionals. The isothermal amplification segment holds around 31% share and is the fastest-growing technology segment, supported by its ability to amplify genetic material at a single, constant temperature without the need for complex thermal cycling equipment, making it ideal for simpler, more portable point-of-care devices.
Why does the infectious disease testing application segment lead the point-of-care molecular diagnostics market?
Based on application, the point-of-care molecular diagnostics market is segregated into infectious disease testing, oncology and cancer screening, cardiology, neurological disorders, and sexual health and sexually transmitted infections. The infectious disease testing segment leads with about 47% of global share, driven by the high burden of infections, the need for rapid pathogen detection, and continued investment in molecular testing after the pandemic. The oncology and cancer screening segment holds around 18% share and is growing at a notable rate as molecular biomarker-based cancer detection enters mainstream clinical practice.
Why does the hospitals and clinics end-user segment lead the point-of-care molecular diagnostics market?
Based on end-user, the point-of-care molecular diagnostics industry is categorized into hospitals and clinics, diagnostic laboratories, ambulatory care centers, home care settings, and pharmacies. The hospitals and clinics segment accounts for approximately 43% of the global market share. It continues to lead the market, driven by the high volume of patient encounters requiring rapid diagnostic decision-making in emergency departments, intensive care units, and outpatient clinics. The home care settings segment holds around 14% share and is growing most rapidly, supported by increasing consumer demand for self-testing solutions and the availability of simple, easy-to-use molecular testing kits that require no clinical training.
How does the direct sales channel dominate the distribution landscape of the point-of-care molecular diagnostics market?
Based on distribution channel, the point-of-care molecular diagnostics market is classified into direct sales, distributors and dealers, and online retail. The direct sales segment accounts for approximately 51% of the global market share and is expected to remain dominant throughout the forecast period, as healthcare institutions purchasing complex molecular testing platforms typically require detailed technical consultation, customized service agreements, regulatory documentation support, and ongoing after-sales training. The online retail segment holds around 19% share and is growing steadily, particularly for consumables, test kits, and accessories, which established users order repeatedly.
Point-Of-Care Molecular Diagnostics Market: Regional Analysis
Why does North America’s strong healthcare, research, and regulations drive its dominance in the point-of-care molecular diagnostics market?
The point-of-care molecular diagnostics market is led by North America, which is expected to grow at a CAGR of about 9.8% and remains the dominant region due to its strong healthcare system and high adoption of advanced diagnostic technologies. The United States is the largest contributor, supported by a high volume of clinical testing, strong insurance coverage for molecular diagnostics, and continuous investment from both government and private companies. The country also has a well-developed research ecosystem, where academic institutions, biotech firms, and diagnostic companies actively work on improving testing technologies. The rapid expansion of testing during the COVID-19 pandemic further strengthened the market by increasing awareness, speeding up regulatory approvals, and encouraging healthcare providers to adopt decentralized testing solutions. Canada also plays an important role with its well-funded public healthcare system and focus on expanding diagnostic services at the community level.
Mexico is gradually emerging as an investment destination, driven by rising private healthcare investment and efforts to improve access in underserved areas. Overall, strong healthcare infrastructure, continuous innovation, and high spending on diagnostics ensure that North America remains the leading region in the point-of-care molecular diagnostics market throughout the forecast period.
What factors make Europe the second-largest region in the point-of-care molecular diagnostics market?
The point-of-care molecular diagnostics market is expected to grow at a CAGR of about 8.7% in Europe, the second-largest region, driven by strong healthcare systems, stringent regulatory standards, and increasing demand for rapid diagnostic testing. The region benefits from clear regulations that ensure diagnostic devices are safe, accurate, and reliable for patient use. Countries across Western and Northern Europe have well-funded public healthcare systems, which create steady demand for fast, advanced diagnostic testing. The European Union has strict rules for in-vitro diagnostic devices, which push companies to develop high-quality molecular testing solutions.
Major countries such as Germany, the United Kingdom, France, the Netherlands, and the Nordic nations play a key role, supported by strong hospital networks, efficient primary care systems, and advanced public health monitoring. These systems rely heavily on quick and accurate diagnostic results to manage diseases effectively. The United Kingdom has increased its focus on molecular diagnostics since the pandemic, investing in genomic research and expanding testing capacity. Europe is also home to several leading manufacturers, particularly in Switzerland, Germany, and the Nordic countries, thereby strengthening local production and supporting continuous innovation.
At the same time, Eastern Europe is becoming more important as healthcare spending rises and access to modern diagnostic technologies improves. Overall, Europe remains a strong and growing market, supported by advanced infrastructure, innovation, and the increasing adoption of point-of-care testing.
Recent Market Developments
- In February 2026, Cepheid was selected by the U.S. Centers for Disease Control and Prevention as a national collaborator to accelerate the development and deployment of rapid point-of-care diagnostic technologies for public health emergencies.
- In November 2025, Abbott Laboratories announced its planned acquisition of Exact Sciences for up to USD 23 billion, strengthening its molecular diagnostics and expanding into cancer testing and screening capabilities.
Point-Of-Care Molecular Diagnostics Market: Competitive Analysis
The leading players in the global point-of-care molecular diagnostics market are:
- Abbott Laboratories
- Roche Diagnostics
- bioMérieux
- Danaher Corporation
- Becton Dickinson and Company
- Hologic Incorporated
- Qiagen
- Meridian Bioscience
- Cepheid (a Danaher company)
- and Chembio Diagnostics
The global point-of-care molecular diagnostics market is segmented as follows:
By Product Type
- Instruments and Analyzers
- Reagents and Consumables
- Software and Services
By Technology
- Polymerase Chain Reaction
- Isothermal Amplification
- Hybridization
- Next-Generation Sequencing
By Application
- Infectious Disease Testing
- Oncology and Cancer Screening
- Cardiology
- Neurological Disorders
- Sexual Health and Sexually Transmitted Infections
By End-User
- Hospitals and Clinics
- Diagnostic Laboratories
- Ambulatory Care Centers
- Home Care Settings
- Pharmacies
By Distribution Channel
- Direct Sales
- Distributors and Dealers
- Online Retail
By Region
- North America
- The U.S.
- Canada
- Mexico
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia Pacific
- The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Table Of Content
Methodology
FrequentlyAsked Questions
Point-of-care molecular diagnostics is a type of medical testing that allows doctors to detect diseases at or near the patient’s location using genetic material, such as DNA or RNA. Instead of sending samples to a distant laboratory and waiting for results, these tests are performed on-site, providing quick results.
- The global point-of-care molecular diagnostics market is projected to grow due to rising infectious and chronic diseases, demand for rapid diagnostics, increasing healthcare investment, advancing affordable technologies, and expanding applications in oncology, pharmacogenomics, and resistance monitoring.
According to a study, the global point-of-care molecular diagnostics market size was worth around USD 8.46 billion in 2024 and is predicted to grow to around USD 17.94 billion by 2034.
The CAGR value of the point-of-care molecular diagnostics market is expected to be around 8.70% during 2025–2034.
North America is expected to lead the global point-of-care molecular diagnostics market during the forecast period, driven by advanced healthcare infrastructure, high diagnostic testing volumes, strong reimbursement frameworks, and the presence of leading global diagnostics companies.
The major players in the global point-of-care molecular diagnostics market include Abbott Laboratories, Roche Diagnostics, bioMerieux, Danaher Corporation, Becton Dickinson and Company, Hologic Incorporated, Qiagen, Meridian Bioscience, Cepheid, and Chembio Diagnostics.
The report examines key aspects of the point-of-care molecular diagnostics market, including a thorough analysis of current growth drivers and restraints, emerging opportunities, significant challenges facing manufacturers and healthcare providers, competitive landscape analysis, detailed regional market breakdowns, and a forward-looking outlook across all major product types, technologies, applications, and geographies.
The point-of-care molecular diagnostics market is expected to see steady growth led by North America, supported by strong healthcare infrastructure and high adoption of advanced diagnostics. Europe follows as the second-largest region, with consistent growth driven by quality standards and the expanding use of rapid testing.
The point-of-care molecular diagnostics market offers opportunities to develop affordable devices, AI-based diagnostic platforms, and home-testing kits. Partnerships among healthcare providers, technology firms, and diagnostic companies can accelerate innovation, expand access, and strengthen global distribution networks.
The point-of-care molecular diagnostics market is shifting toward faster, portable, and easy-to-use tests, with growing preference for home-based testing and digital connectivity for quick results and remote care.
HappyClients


