Rapid Medical Diagnostic Kits Market Size, Share, Trends and Outlook 2032

Rapid Medical Diagnostic Kits Market By Product (OTC Kits and Professional Kits), By Technology (Lateral Flow, Agglutination Assays, Flow-Through, and Solid-phase), By Application (Clinical testing and Home-use), And By Region- Global Industry Perspective, Comprehensive Analysis, and Forecast, 2024-2032
| Market Size in 2023 | Market Forecast in 2032 | CAGR (in %) | Base Year |
|---|---|---|---|
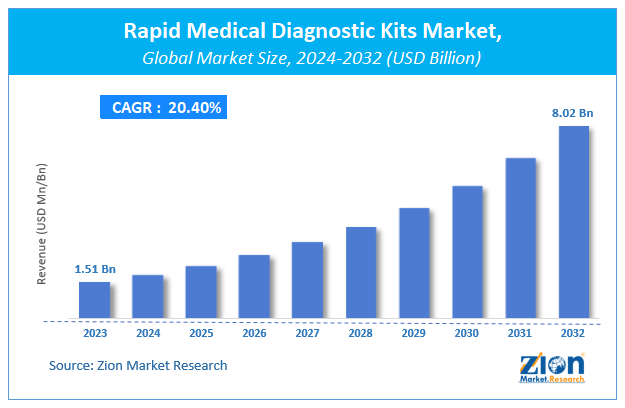
| USD 1.51 Billion | USD 8.02 Billion | 20.4% | 2023 |
Rapid Medical Diagnostic Kits Market Insights
Zion Market Research has published a report on the global Rapid Medical Diagnostic Kits Market, estimating its value at USD 1.51 Billion in 2023, with projections indicating that it will reach USD 8.02 Billion by 2032. The market is expected to expand at a compound annual growth rate (CAGR) of 20.4% over the forecast period 2024-2032. The report explores the factors fueling market growth, the hitches that could hamper this expansion, and the opportunities that may arise in the Rapid Medical Diagnostic Kits Market industry. Additionally, it offers a detailed analysis of how these elements will affect market demand dynamics and market performance throughout the forecast period.
Rapid Medical Diagnostic Kits Market: Synopsis
According to NCBI, rapid medical diagnostic kits have the ability to contain COVID-19 transmission across low as well as middle-income countries across the globe. Apparently, rapid medical diagnostic kits market growth is set to accelerate in the near future subject to a surge in awareness pertaining to the need for effective assessment of chronic ailments, growing point of care diagnostics trends, and massive use of portable rapid testing equipment.
In addition to this, increase in cases of contagious ailments and swift rise in aging population prone to chronic disorders will enhance the popularity of rapid medical diagnostic kits in the healthcare sector. This, in turn, will result in a spike of growth in rapid medical diagnostics kits industry over the forecasting timespan.
Rapid Medical Diagnostic Kits Market: Growth Dynamics
The surge in an aging population prone to chronic disorders will culminate into massive growth of rapid medical diagnostics kits market over years ahead. Apart from this, low costs of products will translate into humongous growth of industry over forthcoming years. Furthermore, the healthcare sector in developing economies is also making use of rapid medical diagnostic kits to facilitate quick disease diagnosis.
This, in turn, will steer growth of rapid medical diagnostic kits industry over upcoming years. With massive increase in disposable income, developed countries have been using rapid medical diagnostics kits since past few years, thereby raising product demand in years ahead. Massive product penetration in the healthcare sector can be attributed to its ability to increase the pace of etiologic diagnosis of patients affected due to chronic disorders.
Furthermore, rise in occurrence of contagious diseases such as COVID-19, Hepatitis-B, Influenza, and Tuberculosis will culminate into immense growth of rapid medical diagnostic kits industry over years ahead. With the introduction of new rapid diagnostics tests for arresting microbial proliferation, the market for rapid medical diagnostics kits is slated to witness elongated growth over years ahead.
Rapid Medical Diagnostic Kits Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Rapid Medical Diagnostic Kits Market |
| Market Size in 2023 | USD 1.51 Billion |
| Market Forecast in 2032 | USD 8.02 Billion |
| Growth Rate | CAGR of 20.4% |
| Number of Pages | 155 |
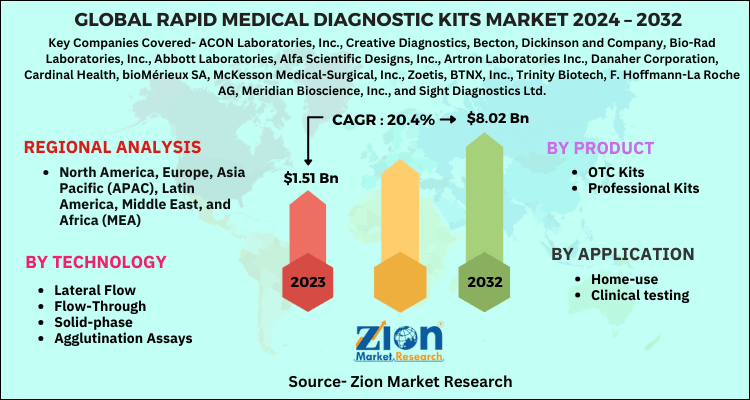
| Key Companies Covered | ACON Laboratories, Inc., Creative Diagnostics, Becton, Dickinson and Company, Bio-Rad Laboratories, Inc., Abbott Laboratories, Alfa Scientific Designs, Inc., Artron Laboratories Inc., Danaher Corporation, Cardinal Health, bioMérieux SA, McKesson Medical-Surgical, Inc., Zoetis, BTNX, Inc., Trinity Biotech, F. Hoffmann-La Roche AG, Meridian Bioscience, Inc., and Sight Diagnostics Ltd. |
| Segments Covered | By Product, By Application, By Technology and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Base Year | 2023 |
| Historical Year | 2018 - 2022 |
| Forecast Year | 2024 - 2032 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Rapid Medical Diagnostic Kits Market: Regional Outlook
North America To Dominate Overall Market Size By 2032
Growth of the regional market over the forecast timespan can be credited to the presence of numerous diagnostic firms in countries such as the U.S. In addition to this, a prominent rise in the number of COVID-19 patients in countries such as the U.S. in the past few years have created immense demand for rapid medical diagnostic kits in the region, thereby driving regional market trends. Additionally, high government focus on preventing disease spread in countries such as Canada and the U.S. has prompted demand for rapid medical diagnostic kits in the region.
Rapid Medical Diagnostic Kits Market: Competitive Landscape
- ACON Laboratories
- Creative Diagnostics
- Becton
- Dickinson and Company
- Bio-Rad Laboratories
- Abbott Laboratories
- Alfa Scientific Designs
- Artron Laboratories
- Danaher Corporation
- Cardinal Health
- bioMérieux SA
- McKesson Medical-Surgical
- Zoetis
- BTNX
- Trinity Biotech
- F. Hoffmann-La Roche AG
- Meridian Bioscience
- and Sight Diagnostics Ltd.
The global Rapid Medical Diagnostic Kits Market is segmented as follows:
By Product
- OTC Kits
- Professional Kits
By Application
- Home-use
- Clinical testing
By Technology
- Lateral Flow
- Flow-Through
- Solid-phase
- Agglutination Assays
By Region
- North America
- The U.S.
- Canada
- Mexico
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia Pacific
- The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Table Of Content
Methodology
FrequentlyAsked Questions
HappyClients