Lipid Nanoparticle Based Genomic Medicines Market Size, Share Report 2034

Lipid Nanoparticle Based Genomic Medicines Market By Product Type (mRNA-Based Systems, siRNA-Based Systems, CRISPR and Gene-Editing Systems and DNA Plasmid-Based Systems), By Application (Oncology, Rare Genetic Disorders, Infectious Diseases and Regenerative Medicine), By End User (Pharmaceutical Companies, Biotechnology Firms, Academic & Research Institutes and Contract Manufacturing Organizations) and By Region - Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, and Forecasts 2025 - 2034
| Market Size in 2024 | Market Forecast in 2034 | CAGR (in %) | Base Year |
|---|---|---|---|
| USD 2.7 Billion | USD 41.7 Billion | 31.5% | 2024 |
Lipid Nanoparticle Based Genomic Medicines Industry Perspective:
What will be the size of the global lipid nanoparticle based genomic medicines market during the forecast period?
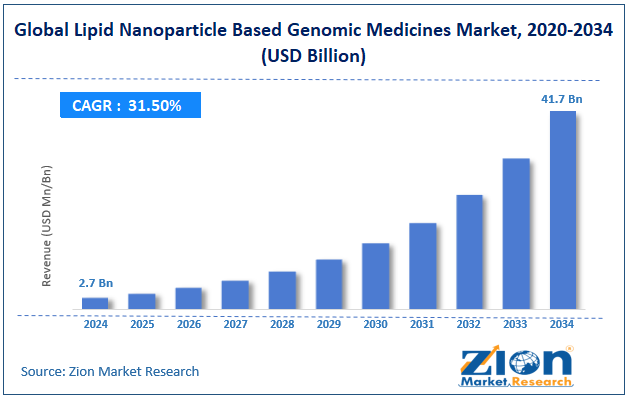
The global lipid nanoparticle based genomic medicines market size was worth around USD 2.7 billion in 2024 and is predicted to grow to around USD 41.7 billion by 2034 with a compound annual growth rate (CAGR) of roughly 31.5% between 2025 and 2034.
 Request Free SampleRequest Free Sample
Request Free SampleRequest Free Sample
Key Insights
- As per the analysis shared by our research analyst, the global lipid nanoparticle based genomic medicines market is estimated to grow annually at a CAGR of around 31.5% over the forecast period (2025-2034).
- In terms of revenue, the global lipid nanoparticle based genomic medicines market size was valued at around USD 2.7 billion in 2024 and is projected to reach USD 41.7 billion by 2034.
- Rising prevalence of chronic & genetic diseases are expected to propel the lipid nanoparticle based genomic medicines market over the projected period.
- Based on the product type, the mRNA-based systems segment held the largest market share of over 60% in 2024.
- Based on the application, the oncology segment captures the largest revenue share of 41% in 2024.
- Based on the end user, the pharmaceutical companies segment dominates the market in 2024 of 38% of revenue share.
- Based on region, North America captures the largest market share of over 55% in 2024.
Lipid Nanoparticle Based Genomic Medicines Market: Overview
Genomic medications based on lipid nanoparticles (LNPs) represent cutting-edge therapies that use lipid-based nanoparticles specifically designed to deliver genetic materials—messenger RNA (mRNA), small interfering RNA (siRNA), DNA, or gene-editing elements—to cells in the body. These nanoparticles act as carriers of the genetic cargo, protecting it from enzyme-mediated breakdown, increasing its stability during circulation in the bloodstream, and promoting delivery into the cell interior. With the help of LNPs, genetic information can be delivered efficiently to target sites and is used for a variety of purposes, ranging from protein replacement therapy to genome editing. As a result, the technology behind LNPs has become particularly popular owing to the success of mRNA vaccines and continues to be widely studied for the treatment of various diseases.
Impact of the USA-Israel War on Iran on the Lipid Nanoparticle Based Genomic Medicines Market
The current war between the USA and Israel over Iran (2026) is having an indirect but substantial effect on the LNP-genomic medicines industry, due to disruptions to the pharmaceutical supply chain, higher input costs, and other logistics challenges. In fact, the war has disrupted trade routes in a dramatic way, especially the Strait of Hormuz, which facilitates the shipment of oil, petrochemicals, and pharmaceutical goods, leading to increased logistics costs, longer delivery times, and the unavailability of key raw materials for pharmaceutical manufacturing.
In addition, the LNP-genomic medicine market relies heavily on specific lipids, nucleic acids, and refrigeration systems during transport, which have increased production costs and made global transportation more difficult. In fact, reports show that medicine prices have gone up, logistics expenses have increased by about 30%, and cancer treatments and insulin drugs are delayed.
Lipid Nanoparticle Based Genomic Medicines Market: Dynamics
Growth Drivers
How does the rapid growth of RNA-based therapeutics drive the lipid nanoparticle based genomic medicines market?
One of the primary factors driving the expansion of the lipid nanoparticle based genomic medicine market is the rapid development of RNA-based therapies, including messenger RNA (mRNA), small interfering RNA (siRNA), and numerous other RNA-based treatments. Indeed, LNP is currently considered the most effective method for delivering such therapies, which makes the market's growth predictable. The growing interest of companies in RNA technology is driving increased investment in delivery systems, such as LNPs. The market's growth is significantly driven by massive corporate investments in the industry. For instance, one of the leading pharmaceutical firms, Bristol Myers Squibb, spent about USD 1.5 billion to purchase Orbital Therapeutics, a company developing an RNA-based therapy delivery system.
Additionally, Novartis invested USD 2.2 billion in a joint venture with Arrowhead Pharmaceuticals for developing RNA-based siRNA therapies. Novo Nordisk also invested USD 550 million into the partnership to produce RNA-based treatments. At the same time, one of the leading mRNA-based therapy producers, Moderna, spends significant sums on infrastructure and research and development. Thus, the company announced its plans to expand mRNA production capacity at a cost of $140 million. Therefore, the above-mentioned stats propel the industry growth.
Restraints
Stringent regulatory & approval challenges are acting as a major barrier to the lipid nanoparticle based genomic medicines industry growth
The strict regulatory approvals and processes pose another significant constraint towards LNP-based genomic medicines. This is because LNP-based genomic medicines are categorized as advanced biologics and gene-based medicines, whose regulatory processes involve several layers of complexity in demonstrating effectiveness, safety profile, distribution, immunogenic potential, and other off-target effects. For instance, the U.S. Food and Drug Administration and the European Medicines Agency have been increasingly raising concerns about the efficacy and safety of gene therapies delivered by LNPs, as these matters are still under study and assessment.
Furthermore, because LNP-based genomic medicines combine lipid and genetic medicines, they are harder to standardize and ensure reproducibility, making the regulatory process much more challenging for developers of the technology. The fact that there are few globally harmonized guidelines for nanomedicine and RNA-based medicines also adds to the difficulties faced during the regulatory approvals. Frequent updates to regulatory expectations, especially regarding novel gene-editing components and lipid chemistries, pose additional hurdles to the development of LNPs.
Opportunities
Why does the rising number of advancements in the sector offer a lucrative opportunity for the lipid nanoparticle based genomic medicines market?
With each passing day, new discoveries and innovations are emerging in the genomic medicine industry, generating a very promising revenue stream for the LNP-based genomic medicines market due to their direct impact on expanding the reach of RNA- and gene-based treatments that use LNPs as carriers. Innovations in mRNA modification, next-generation siRNA, and CRISPR-Cas9 gene editing technology are contributing greatly to expanding the range of diseases that can be treated with RNA and gene-based therapies, including rare genetic syndromes, cancers, and many other illnesses once considered untreatable. On the other hand, ongoing developments in LNP formulation have successfully addressed previous issues related to toxicity, immune responses, and delivery challenges. Additionally, advances in manufacturing technology are enabling reductions in production costs while improving scalability. Moreover, successful clinical trials of RNA-based medications and their regulatory approval are driving investments in the RNA-based drugs industry, which will further stimulate demand for LNP-based genomic medicines.
For instance, in November 2025, Acuitas Therapeutics, a global leader in lipid nanoparticle (LNP) delivery systems for the acceleration of partners’ clinical development, recently presented its Next-Generation LNP advancements, a suite of novel and enhanced technologies that expand the range of diseases treatable with mRNA-LNP medicines, at the 13th International mRNA Health Conference in Berlin. Also at the conference, the company highlighted additional preclinical data on its LNP formulations’ applicability in cancer vaccines, potency, and safety.
Challenges
How does the safety & toxicity concerns pose a significant challenge to the lipid nanoparticle based genomic medicines market?
Safety and toxicity issues have been identified as major challenges for marketing genomic drugs based on lipid nanoparticles (LNPs). The complexity of lipid components' interactions with the nucleic acids they are supposed to transport is considered one of the reasons these problems arise. Thus, specific lipids, such as cationic and ionizable lipids, can trigger an inflammatory response, immunogenicity, and toxicity when the product is administered at high concentrations or frequently. As a result, problems of infusions, entrapment of the drug in the liver, and adverse immune responses can be expected in almost all instances.
As far as safety concerns about lipid particles are concerned, the difficulty is that these considerations pose a problem for regulatory authorities and medical professionals, thereby impeding the drug's commercialization. Moreover, because of the biological characteristics of LNPs, secondary accumulation of nanoparticles can occur in other organs, posing another safety concern related to potential toxicity and undesirable side effects of therapy that require continuous treatment. This problem is complicated by a shortage of long-term clinical research regarding the toxicity, degradation, and influence of nanoparticles on genes and cells. In this context, additional work to develop the drug is necessary and time-consuming.
Lipid Nanoparticle Based Genomic Medicines Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Lipid Nanoparticle Based Genomic Medicines Market |
| Market Size in 2024 | USD 2.7 Billion |
| Market Forecast in 2034 | USD 41.7 Bllion |
| Growth Rate | CAGR of 31.5% |
| Number of Pages | 225 |
| Key Companies Covered | Seed Health, ResBiotic, Fitbiomics, HSK Genetech, Mybacs, ISOThrive, Nouri, Bayer Consumer Health, Clasado Biosciences, CometBio, Essential formulae, ExeGi, Kerry, Immune Biotech, Pendulum Therapeutics, Evolve Biosystems, Gerber, Lovebug probiotics, Mommy's Bliss, Zarbees, Mamasselect, Hyperbiotics, Everidis, Floraster, Enfamil, Culturelle, Biogaia, Viome, Atlas Biomed, Flore, Bioms, Ombre, Biohm, Affinity DNA, Freshly fermented, Carbiotix, Troo, Verisana, Throne, Phable, Biomesight, Invivo, and others. |
| Segments Covered | By Product Type, By Application, By End User, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2034 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Lipid Nanoparticle Based Genomic Medicines Market: Segmentation
Product Type Insights
Why does the mRNA-based systems dominate the lipid nanoparticle based genomic medicines market?
The mRNA-based systems segment held the largest market share of over 60% in 2024. This growth is facilitated by the robust performance of mRNA technology and its natural requirement for an LNP platform. As mRNA treatments are vulnerable to rapid enzymatic breakdown, LNPs play a key role by providing both protection and targeted delivery inside cells, making them vital for this sector. The broad acceptance of mRNA vaccines worldwide, especially those developed by Moderna, Pfizer, and BioNTech, has helped increase revenue substantially. While there is much activity in the use of mRNA to fight infections, promising indications suggest the pipeline will soon expand into cancers (personalized cancer vaccines), genetic diseases, and protein replacements.
Application Insights
Why does the oncology capture the largest market share in the lipid nanoparticle based genomic medicines market?
The oncology segment captures the largest revenue share of 41% in 2024. The rise is driven by the growing use of RNA therapies for cancer treatment and the increasing demand for targeted therapeutic strategies. LNPs provide an important vehicle for delivering mRNA, siRNA, and genome-editing molecules into cancer cells or the immune system. This enables revolutionary treatments such as personalized cancer vaccines, selective tumor targeting with gene-silencing technologies, and modulation of the immune response. Several companies, such as Moderna and BioNTech, are focusing on developing mRNA therapies for cancer. These include novel neoantigen cancer vaccines, which train the body to target its own cancer cells.
End User Insights
Does pharmaceutical companies segment dominate the lipid nanoparticle based genomic medicines market?
The pharmaceutical companies segment dominates the market in 2024 of 38% of revenue share. The increase is attributed to heightened strategic interest in RNA medicines and drug-delivery technologies. Large corporations in the pharma sector have invested in the LNP platform to enhance their pipelines for mRNA vaccines, gene therapy, and RNA interference. Large pharmaceutical organizations, including Pfizer, Moderna, Novartis, and Johnson & Johnson, are well-positioned with robust financial power, manufacturing capabilities, and experience in regulatory affairs. Therefore, they can expedite the clinical advancement of LNP products. Given the success of LNP-based vaccines in the market, pharmaceutical organizations have been encouraged to explore other fields, including cancer, rare diseases, and infectious diseases.
Regional Insights
Why does North America lead the lipid nanoparticle based genomic medicines market?
North America captures the largest market share of over 55% in 2024. The presence of major players and innovative product launch is expected to florish the regional industry. For instance, in March 2026, Hopewell Therapeutics, Inc., a Boston-based next-generation lipid nanoparticle (LNP) company (“Hopewell”), announced that it entered into a sublicense agreement with Foxcroft Therapeutics, Inc., the developer of novel cancer vaccines (“Foxcroft”), granting Foxcroft a global sublicense to certain of its tissue-targeting LNPs (ttLNPs). Hopewell and Foxcroft will collaborate on their evaluation of Hopewell’s ttLNPs as potential delivery vehicles for Foxcroft’s cancer vaccines. Hopewell is currently developing ttLNPs as a delivery vehicle for a broad range of clinical and therapeutic applications, including vaccines, gene therapies, immune-oncology, and pulmonary therapies, targeting tissues in the lung, brain, bone marrow, ovaries, spleen, lymph nodes, kidneys, liver, and other vital organs. Pre-clinical testing has demonstrated Hopewell ttLNPs to perform more effectively than other LNPs available in the market.
Lipid Nanoparticle Based Genomic Medicines Market: Competitive Analysis
The global lipid nanoparticle based genomic medicines market is dominated by players like:
- Seed Health
- ResBiotic
- Fitbiomics
- HSK Genetech
- Mybacs
- ISOThrive
- Nouri
- Bayer Consumer Health
- Clasado Biosciences
- CometBio
- Essential formulae
- ExeGi
- Kerry
- Immune Biotech
- Pendulum Therapeutics
- Evolve Biosystems
- Gerber
- Lovebug probiotics
- Mommy's Bliss
- Zarbees
- Mamasselect
- Hyperbiotics
- Everidis
- Floraster
- Enfamil
- Culturelle
- Biogaia
- Viome
- Atlas Biomed
- Flore
- Bioms
- Ombre
- Biohm
- Affinity DNA
- Freshly fermented
- Carbiotix
- Troo
- Verisana
- Throne
- Phable
- Biomesight
- Invivo
The global lipid nanoparticle based genomic medicines market is segmented as follows:
By Product Type
- mRNA-Based Systems
- siRNA-Based Systems
- CRISPR and Gene-Editing Systems
- DNA Plasmid-Based Systems
By Application
- Oncology
- Rare Genetic Disorders
- Infectious Diseases
- Regenerative Medicine
By End User
- Pharmaceutical Companies
- Biotechnology Firms
- Academic & Research Institutes
- Contract Manufacturing Organizations
By Region
- North America
- The U.S.
- Canada
- Mexico
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia Pacific
- The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Table Of Content
Methodology
FrequentlyAsked Questions
Genomic medications based on lipid nanoparticles (LNPs) represent cutting-edge therapies that use lipid-based nanoparticles specifically designed to deliver genetic materials—messenger RNA (mRNA), small interfering RNA (siRNA), DNA, or gene-editing elements—to cells in the body.
Key growth drivers include the rapid expansion of RNA-based therapeutics, increasing demand for precision medicine, advancements in nanotechnology, and rising investments by pharmaceutical companies in LNP-enabled drug delivery systems.
Major challenges include safety and toxicity concerns, stringent regulatory requirements, high development and manufacturing costs, and complexities in large-scale production and targeted delivery.
Based on the product type, the mRNA-based systems segment is expected to dominate the lipid nanoparticle based genomic medicines market growth during the projected period.
Emerging trends include next-generation LNP formulations, targeted and organ-specific delivery, integration with CRISPR-Cas9 and RNA therapies, scalable manufacturing innovations, and expanding applications in personalized medicine and oncology.
According to the report, the global lipid nanoparticle based genomic medicines market size was worth around USD 2.7 billion in 2024 and is predicted to grow to around USD 41.7 billion by 2034.
The global lipid nanoparticle based genomic medicines market is expected to grow at a CAGR of 31.5% during the forecast period.
The global lipid nanoparticle based genomic medicines industry growth is expected to be led by North America over the forecast period.
Which are the major players leveraging the lipid nanoparticle based genomic medicines market growth?
The global lipid nanoparticle based genomic medicines market is dominated by players like Seed Health, ResBiotic, Fitbiomics, HSK Genetech, Mybacs, ISOThrive, Nouri, Bayer Consumer Health, Clasado Biosciences, CometBio, Essential formulae, ExeGi, Kerry, Immune Biotech, Pendulum Therapeutics, Evolve Biosystems, Gerber, Lovebug probiotics, Mommy's Bliss, Zarbees, Mamasselect, Hyperbiotics, Everidis, Floraster, Enfamil, Culturelle, Biogaia, Viome, Atlas Biomed, Flore, Bioms, Ombre, Biohm, Affinity DNA, Freshly fermented, Carbiotix, Troo, Verisana, Throne, Phable, Biomesight and Invivo among others.
The market report covers the geographical market along with a comprehensive competitive landscape analysis. It also includes cash flow analysis, profit ratio analysis, market basket analysis, market attractiveness analysis, sentiment analysis, PESTLE analysis, trend analysis, SWOT analysis, trade area analysis, demand & supply analysis, Porter’s five forces analysis, and value chain analysis.
HappyClients


