Beclomethasone Dipropionate Market Size, Share, Growth and Forecast 2034

Beclomethasone Dipropionate Market By Formulation Type (Inhalers, Nasal Sprays, Topical Creams, Ointments, Lotions, and Others), By Application (Asthma Treatment, Allergic Rhinitis, Dermatological Conditions, Chronic Obstructive Pulmonary Disease, Inflammatory Bowel Disease, Other Respiratory Conditions), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Clinics), By End-User (Hospitals, Clinics, Homecare Settings, Ambulatory Surgical Centers), and By Region - Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, and Forecasts 2025 - 2034
| Market Size in 2024 | Market Forecast in 2034 | CAGR (in %) | Base Year |
|---|---|---|---|
| USD 1207.64 Million | USD 2228.45 Million | 5.82% | 2024 |
Beclomethasone Dipropionate Industry Prospective
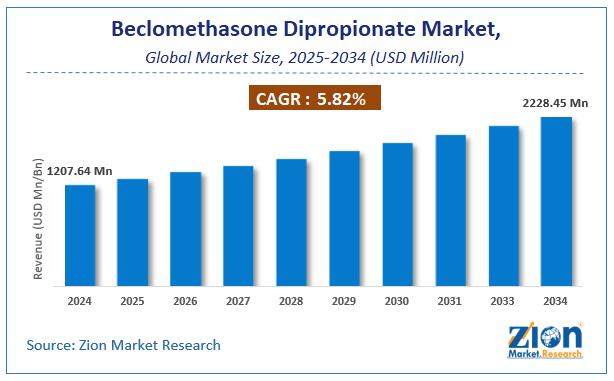
The global beclomethasone dipropionate market size was worth approximately USD 1207.64 Million in 2024 and is projected to grow to around USD 2228.45 Million by 2034, with a compound annual growth rate (CAGR) of roughly 5.82% between 2025 and 2034.
 Request Free SampleRequest Free Sample
Request Free SampleRequest Free Sample
Key Insights
- As per the analysis shared by our research analyst, the global beclomethasone dipropionate market is estimated to grow annually at a CAGR of around 5.82% over the forecast period (2025-2034).
- In terms of revenue, the global beclomethasone dipropionate market size was valued at approximately USD 1207.64 Million in 2024 and is projected to reach USD 2228.45 Million by 2034.
- The beclomethasone dipropionate market is projected to grow significantly due to the increasing prevalence of asthma and allergic conditions, growing awareness about respiratory health, and rising adoption of corticosteroid therapy.
- Based on formulation type, the inhalers segment is expected to lead the beclomethasone dipropionate market, while the nasal sprays segment is anticipated to experience significant growth.
- Based on application, the asthma treatment segment is expected to lead the beclomethasone dipropionate market, while the allergic rhinitis segment is anticipated to witness notable growth.
- Based on distribution channel, the hospital pharmacies segment is the dominating segment, while the online pharmacies segment is projected to witness sizeable revenue over the forecast period.
- Based on end-user, the hospitals segment is expected to lead the market compared to the home care settings segment.
- Based on region, North America is projected to dominate the global beclomethasone dipropionate market during the estimated period, followed by Europe.
Beclomethasone Dipropionate Market: Overview
Beclomethasone dipropionate is a corticosteroid medicine used to control inflammation and swelling in the body, especially in the airways, nose, skin, and digestive system. It works by reducing immune activity and lowering the release of inflammatory mediators, thereby helping to prevent irritation and tissue damage. Doctors commonly prescribe beclomethasone dipropionate for asthma treatment, where regular use supports better breathing control and lowers the risk of asthma attacks. It is also widely used for allergic rhinitis, helping relieve nasal congestion, sneezing, itching, and a runny nose caused by pollen, dust, or other allergens.
In dermatology, beclomethasone dipropionate creams and ointments are used to manage eczema, psoriasis, and dermatitis by reducing redness, swelling, and skin discomfort. The medicine is available as inhalers, nasal sprays, and topical formulations to suit different medical needs. Healthcare professionals prefer this anti-inflammatory drug because it provides strong local action with fewer whole-body side effects compared to oral steroids. Beclomethasone dipropionate supports long-term disease control, improves symptom stability, and enhances overall quality of life.
The rising prevalence of respiratory diseases and allergic conditions worldwide is expected to drive growth in the beclomethasone dipropionate market throughout the forecast period.
Beclomethasone Dipropionate Market Dynamics
Growth Drivers
How is the increasing prevalence of respiratory diseases driving the beclomethasone dipropionate market growth?
The beclomethasone dipropionate market shows strong growth as global respiratory diseases increase due to pollution exposure, urban living, and long-term lifestyle changes. Air pollution from vehicle and industrial emissions, as well as from fossil fuel combustion, releases harmful particles, gases, and allergens that irritate the airways and increase asthma prevalence. Climate change lengthens pollen seasons, raises pollen levels, and supports mold growth, leading to higher allergic rhinitis rates and chronic breathing discomfort. Poor indoor air quality caused by limited ventilation, tobacco smoke, and chemical exposure worsens lung health across homes, workplaces, and public buildings. Rising childhood asthma cases, aging populations, obesity, and low physical activity increase long-term dependence on inhaled corticosteroid therapies worldwide. Growing awareness of respiratory care after recent global health events further supports demand for effective asthma treatments and anti-inflammatory medications.
Advancements in drug delivery systems and formulation technology
The global beclomethasone dipropionate industry benefits from steady pharmaceutical technology innovation, improving drug delivery efficiency, patient comfort, treatment reliability, and inhaled corticosteroid therapy outcomes worldwide. Modern metered dose inhalers use breath-activated designs coordinating medication release with inhalation, improving lung deposition and reducing oral drug loss. Dry powder inhalers remove propellants and deliver consistent doses, reducing coordination challenges for asthma patients using long-term respiratory treatment. Advanced small particle formulations reach deeper lung areas, improving control of airway inflammation linked with chronic asthma and allergic respiratory disorders.
Nasal spray systems offer precise dose control, supporting reliable allergic rhinitis treatment and consistent symptom relief during long pollen exposure periods. Environment-friendly hydrofluoroalkane propellants replace older chemicals, supporting sustainable inhaler production and compliance with global environmental safety regulations. Smart inhalers with sensors support medication adherence by tracking usage patterns, sending reminders, and improving long-term asthma management outcomes. Extended release formulations, combination therapies, ergonomic designs, and built-in dose counters improve treatment convenience, accessibility, and confidence for patients across all age groups.
Restraints
Side effects and safety concerns are limiting widespread adoption
The beclomethasone dipropionate market faces challenges due to treatment side effects, reducing patient confidence, and limiting prescribing for sensitive patient groups. Long-term inhaled corticosteroid use may cause oral thrush, hoarseness, and voice changes due to medication residue affecting the mouth, throat, and vocal cords. Extended or high-dose exposure allows systemic absorption, leading to adrenal suppression, slowed growth, and hormonal imbalance concerns. Topical beclomethasone dipropionate use may result in skin thinning, stretch marks, bruising, and eye issues such as cataracts or glaucoma.
Chronic corticosteroid therapy increases infection risk, lowers bone density, and raises osteoporosis, fracture, and metabolic health concerns in older adults. Weight gain, blood sugar changes, mood disturbance, anxiety, and sleep problems reduce acceptance of long-term asthma and allergy treatment. Incorrect inhaler technique, safety worries, and dependency fears reduce adherence, limiting the beclomethasone dipropionate market growth despite proven therapeutic benefits.
Opportunities
How is the growing demand for generic medications creating opportunities for the beclomethasone dipropionate industry?
The beclomethasone dipropionate industry shows strong growth as patent expiries and healthcare cost pressures increase demand for affordable generic respiratory medicines. Generic beclomethasone dipropionate products deliver equal asthma control and anti-inflammatory benefits while offering lower prices for wider patient access. Hospitals, insurers, and public health systems encourage generic prescribing to reduce treatment costs while maintaining quality inhaled corticosteroid therapy. Emerging economies benefit from the availability of generic respiratory medications, thereby improving access to asthma care across urban and rural populations, regions, and healthcare settings nationwide.
Generic drug manufacturers invest in improved formulations, delivery devices, and combination inhalers supporting adherence, convenience, and competitive market positioning. Simplified regulatory approval pathways, faster market entry, and expanding online pharmacy channels strengthen global generic beclomethasone dipropionate distribution. Government initiatives, patient assistance programs, and middle-class expansion increase acceptance of affordable inhaled corticosteroids in developing pharmaceutical markets. Higher quality standards, strategic partnerships, and supply reliability improve physician confidence, supporting sustained generic beclomethasone dipropionate market expansion.
Challenges
How are regulatory complexities and approval processes creating obstacles for the beclomethasone dipropionate market?
The beclomethasone dipropionate industry faces major challenges from strict regulatory requirements, increasing development costs, delayed approvals, and slowed new respiratory product launches worldwide. Health authorities require extensive clinical trials proving safety, efficacy, and consistent drug delivery before approving new inhalers, formulations, or corticosteroid delivery technologies. Generic beclomethasone dipropionate producers must conduct bioequivalence studies demonstrating equivalent absorption, dose performance, and therapeutic outcomes compared with reference-branded medicines. Different regulatory standards across regions require separate approval pathways, raising costs, timelines, documentation effort, and operational complexity for global pharmaceutical manufacturers.
Manufacturers face added pressure from environmental inhaler rules, quality audits, inspections, pricing controls, and pharmacovigilance reporting requirements. Intellectual property disputes, manufacturing validation, stability testing, multilingual labeling, and child-safe packaging requirements increase compliance risk, cost, and administrative workload. Frequent regulatory updates, guidance documents, and environmental impact assessments create uncertainty that affects investment decisions, the pace of innovation, and the growth of the beclomethasone dipropionate market.
 Request Free SampleRequest Free Sample
Request Free SampleRequest Free Sample
Beclomethasone Dipropionate Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Beclomethasone Dipropionate Market |
| Market Size in 2024 | USD 1207.64 Million |
| Market Forecast in 2034 | USD 2228.45 Million |
| Growth Rate | CAGR of 5.82% |
| Number of Pages | 260 |
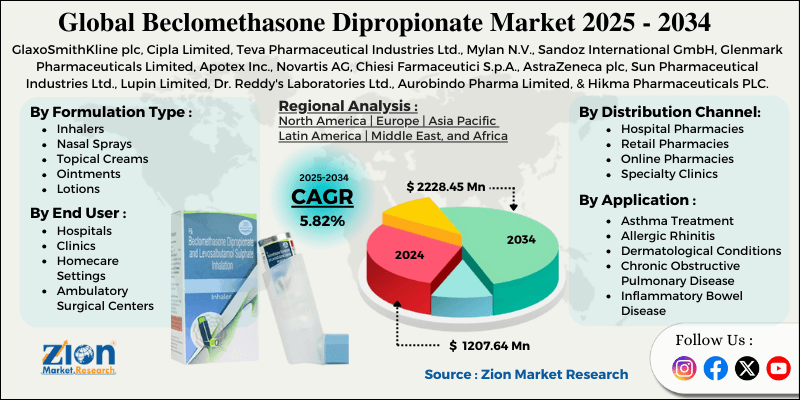
| Key Companies Covered | GlaxoSmithKline plc, Cipla Limited, Teva Pharmaceutical Industries Ltd., Mylan N.V., Sandoz International GmbH, Glenmark Pharmaceuticals Limited, Apotex Inc., Novartis AG, Chiesi Farmaceutici S.p.A., AstraZeneca plc, Sun Pharmaceutical Industries Ltd., Lupin Limited, Dr. Reddy's Laboratories Ltd., Aurobindo Pharma Limited, and Hikma Pharmaceuticals PLC. |
| Segments Covered | By Formulation Type, By Application, By Distribution Channel, By End User and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America,The Middle East and Africa (MEA) |
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2034 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Beclomethasone Dipropionate Market: Segmentation
The global beclomethasone dipropionate market is segmented based on formulation type, application, distribution channel, end-user, and region.
Based on formulation type, the global beclomethasone dipropionate industry is divided into inhalers, nasal sprays, topical creams, ointments, lotions, and others. Inhalers lead the market due to their effectiveness in delivering medication directly to the lungs, high patient compliance compared to oral medications, and widespread physician preference for managing respiratory conditions.
Based on application, the industry is categorized into asthma treatment, allergic rhinitis, dermatological conditions, chronic obstructive pulmonary disease, inflammatory bowel disease, and other respiratory conditions. Asthma treatment leads the market due to the high global prevalence of asthma and the proven effectiveness of inhaled corticosteroids in disease management.
Based on distribution channel, the global beclomethasone dipropionate market is classified into hospital pharmacies, retail pharmacies, online pharmacies, and specialty clinics. Hospital pharmacies are expected to lead the market during the forecast period, owing to their role in the initial diagnosis and treatment of severe respiratory conditions in hospital settings and to their integration with electronic prescribing systems.
Based on end-user, the global market is segregated into hospitals, clinics, home care settings, and ambulatory surgical centers. Hospitals hold the largest market share due to their role in treating acute exacerbations, providing comprehensive diagnostic services, facilitating access to specialist physicians, and managing complex cases requiring multidisciplinary care.
Beclomethasone Dipropionate Market: Regional Analysis
What factors are contributing to North America's dominance in the global beclomethasone dipropionate market?
North America accounted for an estimated 38% share of the global beclomethasone dipropionate market, maintaining its position as the leading regional market during the forecast period. North America leads the beclomethasone dipropionate market due to high asthma prevalence, healthcare systems, insurance coverage, and respiratory disease awareness. The United States reports millions of children and adults requiring inhaled corticosteroid therapy for asthma control and allergy management. Well-developed healthcare infrastructure supports early diagnosis, routine monitoring, and long-term management of chronic respiratory conditions across the region.
Public and private insurance programs reduce treatment costs, improving access to prescribed beclomethasone dipropionate inhalers and nasal sprays. Medical professionals follow updated clinical guidelines promoting inhaled corticosteroids as first-line therapy for persistent asthma symptoms globally. Efficient pharmaceutical distribution networks ensure the steady availability of respiratory medicines across hospitals, clinics, retail pharmacies, and specialty care centers. Patient education initiatives and advocacy groups raise awareness of asthma triggers, medication adherence, and respiratory health. Advanced air quality monitoring, pollen forecasts, and health tools help patients manage symptoms and optimize preventive inhaler use. Leading research institutions conduct clinical studies to improve understanding of the effectiveness and safety profiles of corticosteroids and to optimize dosing strategies.
Strong intellectual property frameworks encourage pharmaceutical investment, innovation, and development of beclomethasone dipropionate formulations. Clear regulatory pathways from agencies such as the FDA support timely approval while maintaining high safety standards nationwide. Widespread use of spirometry diagnostic testing enables accurate asthma diagnosis, treatment adjustment, and ongoing patient outcome monitoring. Telemedicine growth and specialty pharmacy services improve access, education, adherence support, and continuity of care for respiratory patients. Strong reimbursement policies support consistent medication use, reducing emergency visits, hospitalizations, and asthma-related healthcare burden.
Europe is maintaining a strong market presence.
Europe held an estimated 27% share of the global beclomethasone dipropionate market, positioning it as the second-leading region, supported by strong healthcare systems and high generic adoption. Europe holds a strong position in the beclomethasone dipropionate market, supported by universal healthcare systems, environmental health focus, and structured pharmaceutical regulation frameworks. Major countries, including Germany, France, and the United Kingdom, operate advanced healthcare infrastructure, ensuring steady access to essential respiratory medications. Universal healthcare coverage lowers financial burden, allowing more patients to receive inhaled corticosteroid treatment for asthma and allergic conditions.
Strict air quality standards and environmental protection policies increase public awareness of respiratory health risks across urban populations. National health services negotiate bulk medicine purchases, supporting affordable pricing while maintaining consistent quality and reliable supply chains. The European Medicines Agency offers harmonized regulatory oversight, simplifying approval processes across multiple regional pharmaceutical markets. Generic beclomethasone dipropionate adoption remains high due to government policies supporting cost-effective respiratory treatment strategies. Comprehensive patient registries and epidemiological studies provide accurate data on asthma prevalence, treatment outcomes, and healthcare planning.
Pharmaceutical companies maintain strong research and development operations, driving innovation in inhaler technology, formulation, and drug delivery systems. Public health authorities highlight climate change respiratory impacts, encouraging preventive care and long-term disease management approaches. Smoking cessation programs and strict tobacco control policies reduce respiratory disease burden and support improved lung health outcomes. Medical education programs train healthcare professionals in standardized, evidence-based practices for the diagnosis, monitoring, and treatment of respiratory diseases. Cross-border healthcare agreements allow patient mobility, improving access to specialized respiratory care across European healthcare systems.
Recent Market Developments
- In October 2025, Amneal Pharmaceuticals announced that the FDA’s tentative approval of its ANDA for beclomethasone dipropionate HFA inhalation aerosol represents the company’s first metered-dose inhaler product, marking an expansion into complex respiratory therapies.
Beclomethasone Dipropionate Market: Competitive Analysis
The leading players in the global beclomethasone dipropionate market are -
- GlaxoSmithKline plc
- Cipla Limited
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V.
- Sandoz International GmbH
- Glenmark Pharmaceuticals Limited
- Apotex Inc.
- Novartis AG
- Chiesi Farmaceutici S.p.A.
- AstraZeneca plc
- Sun Pharmaceutical Industries Ltd.
- Lupin Limited
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Limited
- Hikma Pharmaceuticals PLC.
The global beclomethasone dipropionate market is segmented as follows:
By Formulation Type
- Inhalers
- Nasal Sprays
- Topical Creams
- Ointments
- Lotions
- Others
By Application
- Asthma Treatment
- Allergic Rhinitis
- Dermatological Conditions
- Chronic Obstructive Pulmonary Disease
- Inflammatory Bowel Disease
- Other Respiratory Conditions
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Specialty Clinics
By End User
- Hospitals
- Clinics
- Homecare Settings
- Ambulatory Surgical Centers
By Region
- North America
- The U.S.
- Canada
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Table Of Content
Methodology
FrequentlyAsked Questions
HappyClients


