Global Medical Device Complaint Management Market Size, Share, Growth Analysis Report - Forecast 2034

Medical Device Complaint Management Market By Service (Complaints Log/Intake, Product Surveillance & Regulatory Compliance, Returned/Non-Returned Product Analysis and Resolve & Closure), By Application (Small & Medium Enterprise and Large Enterprise), and By Region: Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, and Forecasts 2025 - 2034
| Market Size in 2024 | Market Forecast in 2034 | CAGR (in %) | Base Year |
|---|---|---|---|
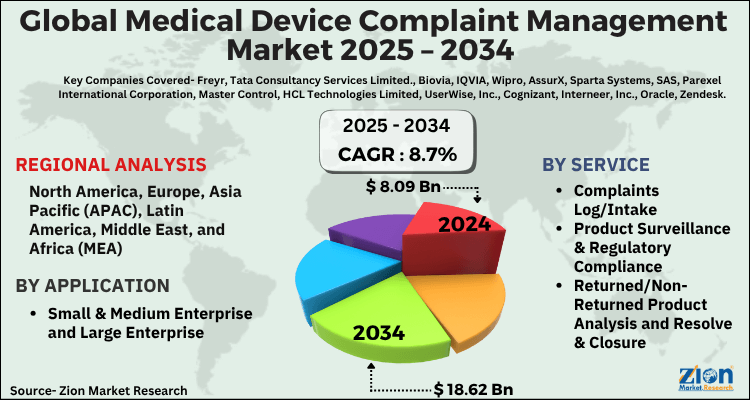
| USD 8.09 Billion | USD 18.62 Billion | 8.7% | 2024 |
Global Medical Device Complaint Management Market: Industry Perspective
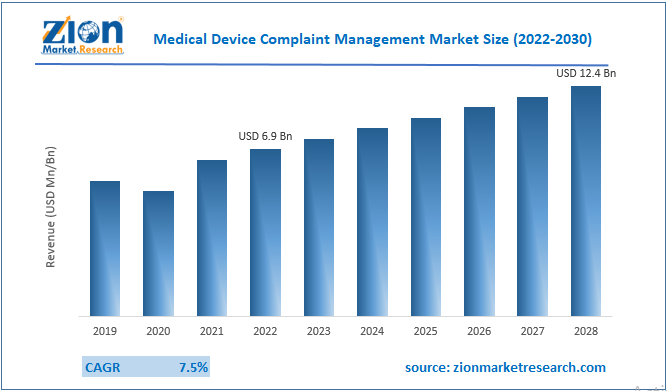
The global medical device complaint management market size was worth around USD 8.09 Billion in 2024 and is predicted to grow to around USD 18.62 Billion by 2034 with a compound annual growth rate (CAGR) of roughly 8.7% between 2025 and 2034. The report analyzes the global medical device complaint management market's drivers, restraints/challenges, and the effect they have on the demands during the projection period. In addition, the report explores emerging opportunities in the medical device complaint management industry.
The report analyzes the global medical device complaint management market’s drivers, restraints/challenges, and the effect they have on the demands during the projection period. In addition, the report explores emerging opportunities in the medical device complaint management industry.
Global Medical Device Complaint Management Market: Overview
Within a quality management system (QMS), complaint management software plays a crucial role in enabling manufacturers to promptly process concerns and, where necessary, involve suppliers. To support continuous improvement, complaint management software also offers feedback on product-in-use data to the design, planning, and process engineering teams of the manufacturer and/or supplier. The manufacturer's complaint gathering, analysis, and evaluation processes are made possible by the usage of a QMS concern and complaint management (CCM) software module. The evaluations that follow and vulnerability studies are crucial to the advancement of products and procedures. Software for managing complaints and concerns also enables manufacturers to streamline and synchronize supply chain operations with their integrated processes. Continuous improvement is made possible by CCM software, which includes internal problem-solving procedures as well as communication between companies and suppliers.
Global Medical Device Complaint Management Market: Growth Drivers
The growing number of safety incidents associated with the use of medical devices drives the market growth
The number of medical device reports received by the US Food and Drug Administration (FDA) increased significantly between 2020 and 2021, according to the agency. When compared to 2020, when the FDA received 1,225,110 medical devices, the number received in 2021 was roughly 1,463,210, a 19.6% increase. The necessity to record any events involving the use of medical equipment and the growing awareness of patient safety are both responsible for this increase. This, in turn, is anticipated to boost the global medical device complaint management market demand.
Restraints:
Improper classification of medical device complaints impedes the market growth
An endless cycle of escalating issues might result from improperly categorizing complaints. It distorts trending, tracking, and w. Regulators specifically check for it (and frequently issue observations for it) since it may lead to MDRs not being filed when they should be. In addition to causing backlogs, improper classification can make it more difficult to clear them by requiring the time-consuming process of re-opening and re-investigating closed files.
Opportunities:
Innovative product launch by the key player provides a lucrative opportunity
The medical device complaint management market's key players are always working to develop novel product development strategies to obtain a competitive edge and meet the market's increasing demand. For instance, major firms are creating cutting-edge mobile application solutions to improve the management of client complaints. For instance, Intellect provides a platform for integrated mobile applications that users can use to file complaints, escalate issues, and create queries. The Intellect mobile app is conveniently offered on the App Store and Google Play Store, where it may be downloaded as an Intellect app or a mobile app with a different brand.
Challenges:
Inadequate training for risk assignment and ranking poses a major challenge
The severity of each complaint should be rated, then it should be prioritized. However, a lot of complaint handlers lack the training necessary to do the amount of analysis needed for this job. A product engineer with extensive knowledge of both how to utilize the software that assigns risk numbers and the factors the program is considering to arrive at its response is often needed to assign risk effectively and consistently. Thus, inadequate training for risk assignment and ranking is expected to pose a major challenge to the medical device complaint management market growth over the forecast period.
Key Insights
- As per the analysis shared by our research analyst, the global medical device complaint management market is estimated to grow annually at a CAGR of around 8.7% over the forecast period (2025-2034).
- Regarding revenue, the global medical device complaint management market size was valued at around USD 8.09 Billion in 2024 and is projected to reach USD 18.62 Billion by 2034.
- The medical device complaint management market is projected to grow at a significant rate due to stringent regulatory compliance requirements and increasing complexity in medical device post-market surveillance.
- Based on Service, the Complaints Log/Intake segment is expected to lead the global market.
- On the basis of Application, the Small & Medium Enterprise and Large Enterprise segment is growing at a high rate and will continue to dominate the global market.
- Based on region, North America is predicted to dominate the global market during the forecast period.
Global Medical Device Complaint Management Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Medical Device Complaint Management Market |
| Market Size in 2024 | USD 8.09 Billion |
| Market Forecast in 2034 | USD 18.62 Billion |
| Growth Rate | CAGR of 8.7% |
| Number of Pages | 211 |
| Key Companies Covered | Freyr, Tata Consultancy Services Limited., Biovia, IQVIA, Wipro, AssurX, Sparta Systems, SAS, Parexel International Corporation, Master Control, HCL Technologies Limited, UserWise, Inc., Cognizant, Interneer, Inc., Oracle, Zendesk, Thryv, Inc, BizzMine, PTC, Orcanos, ArisGlobal LLC and AmpleLogic among others., and others. |
| Segments Covered | By Service, By Application, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, The Middle East and Africa (MEA) |
| Base Year | 2024 |
| Historical Year | 2020 to 2023 |
| Forecast Year | 2025 - 2034 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Global Medical Device Complaint Management Market: Segmentation
The global medical device complaint management industry is segmented based on service type and region. All the segments have been analyzed based on present and future trends and the market is estimated from 2025 to 2034.
Based on the service type, the global market is bifurcated into complaints log/intake, product surveillance & regulatory compliance, returned/ non-returned product analysis, and resolve & closure. Product surveillance & regulatory compliance is expected to dominate the market over the forecast period. Because adverse events, product flaws, and medical device failures cause pain, injury, or even death, regulatory bodies, and consumers are becoming less tolerant. Furthermore, to properly track product performance and compliance, regulatory bodies continually emphasize the value of product surveillance.
The medical device vigilance/medical device reporting segment is anticipated to experience the fastest growth rate during the next years within the product surveillance segment. Regulations reforms carried out by regulatory bodies like the U.S. FDA are responsible for this lucrative expansion. For instance, the U.S. FDA has mandated that businesses submit to it immediately any concerns about adverse events, product defects, or failures relating to medical devices. These required regulatory improvements will come with severe penalties for non-compliance.
On the other hand, the complaints log/intake is expected to grow significantly during the forecast period. This section is a gateway mechanism for preventative or corrective action as well as post-market actions. It is the initial phase of complaint handling. Since the complaint must be evaluated to see if it qualifies as a reportable adverse event, complaint files are connected to the medical device reporting (MDR) event file. Additionally, it is required that medical device manufacturers record and monitor complaints by the quality system regulation (QSR), which is outlined in 21 CFR Part 820. Thus, this is expected to drive segment growth over the forecast period.
Recent Developments:
- In June 2024, InfectoPharm Arzneimittel und Consilium GmbH, the top pediatric pharmaceutical manufacturer in Germany, has chosen TrackWise Digital® as its end-to-end, next-generation quality management platform, according to a news release from Sparta Systems, a Honeywell company. To seamlessly integrate quality processes and data across its manufacturing operations and suppliers, InfectoPharm will implement TrackWise Digital's solution suite, which includes core QMS processes in addition to Complaints Handling, Supplier Quality Management, Document Management, and Training Management.
- In November 2024, a new module of IQVIA's range of Orchestrated Customer Engagement (OCE) products, Grants, and funding administration, has been made available. This initiative is anticipated to give the life sciences sector a way to effectively administer and supervise their international strategic giving programs.
Global Medical Device Complaint Management Market: Regional Analysis
The Medical Device Complaint Management Market exhibits significant regional variation, with North America holding a dominant position due to its well-established regulatory framework, high adoption of advanced healthcare technologies, and the strong presence of key medical device manufacturers. Europe follows closely, driven by stringent compliance requirements and a growing emphasis on patient safety and post-market surveillance. The Asia-Pacific region is expected to witness the fastest growth, propelled by expanding healthcare infrastructure, increasing medical device production, and rising awareness of regulatory standards across emerging economies such as China and India. Meanwhile, Latin America, the Middle East, and Africa are gradually adopting complaint management systems, supported by regulatory improvements and a growing focus on quality assurance in healthcare delivery.
Global Medical Device Complaint Management Market: Competitive Analysis
The report provides a company market share analysis to give a broader overview of the key market players. In addition, the report also covers key strategic developments of the market, including acquisitions & mergers, new product launches, agreements, partnerships, collaborations & joint ventures, research & development, and regional expansion of major participants involved in the medical device complaint management market on a global and regional basis.
The global medical device complaint management market is dominated by players like:
- Freyr
- Tata Consultancy Services Limited.
- Biovia
- IQVIA
- Wipro
- AssurX
- Sparta Systems
- SAS
- Parexel International Corporation
- Master Control
- HCL Technologies Limited
- UserWise Inc.
- Cognizant
- Interneer Inc.
- Oracle
- Zendesk
- Thryv Inc.
- BizzMine
- PTC
- Orcanos
- ArisGlobal LLC and AmpleLogic among others.
Global Medical Device Complaint Management Market: Segmentation Analysis
The global medical device complaint management market is segmented as follows;
By Service
- Complaints Log/Intake
- Product Surveillance & Regulatory Compliance
- Returned/Non-Returned Product Analysis and Resolve & Closure
By Application
- Small & Medium Enterprise and Large Enterprise
Global Medical Device Complaint Management Market: Regional Segment Analysis
- North America
- The U.S.
- Canada
- Mexico
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia Pacific
- The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Table Of Content
Methodology
FrequentlyAsked Questions
Incorporating software helps organizations in maintaining better documentation. A multi-page electronic form ensures precise recording of complaint data along with reorganizing documentation into a simple process. This helps in maintaining transparency and improves decision-making related to customer complaints. The demand and popularity of the medical device complaint management market across the globe can be attributed to the growing software used to support the overall process of complaint management.
The global medical device complaint management market is expected to grow due to stringent regulatory compliance requirements and increasing complexity in medical device post-market surveillance.
According to a study, the global medical device complaint management market size was worth around USD 8.09 Billion in 2024 and is expected to reach USD 18.62 Billion by 2034.
The global medical device complaint management market is expected to grow at a CAGR of 8.7% during the forecast period.
North America is expected to dominate the medical device complaint management market over the forecast period.
Leading players in the global medical device complaint management market include Freyr, Tata Consultancy Services Limited., Biovia, IQVIA, Wipro, AssurX, Sparta Systems, SAS, Parexel International Corporation, Master Control, HCL Technologies Limited, UserWise, Inc., Cognizant, Interneer, Inc., Oracle, Zendesk, Thryv, Inc, BizzMine, PTC, Orcanos, ArisGlobal LLC and AmpleLogic among others., among others.
The report explores crucial aspects of the medical device complaint management market, including a detailed discussion of existing growth factors and restraints, while also examining future growth opportunities and challenges that impact the market.
HappyClients