Global Medical Inhaled Nitric Oxide Market Size, Share, Growth Analysis Report - Forecast 2034

Medical Inhaled Nitric Oxide Market By Product Type (Inhaled Nitric Oxide Delivery Systems, Nitric Oxide Sensors and Monitors), By Route of Administration (Inhaled Nitric Oxide Gas, Inhaled Nitric Oxide with Oxygen Mixtures), By Application (Cardiovascular Disorders, Respiratory Disorder), By End User (Hospitals, Clinics), and By Region: Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, and Forecasts 2025 - 2034
| Market Size in 2024 | Market Forecast in 2034 | CAGR (in %) | Base Year |
|---|---|---|---|
| USD 862.5 Million | USD 1846.95 Million | 7.3% | 2024 |
Medical Inhaled Nitric Oxide Market: Industry Perspective
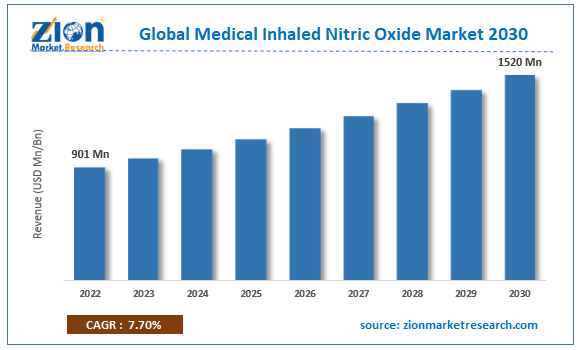
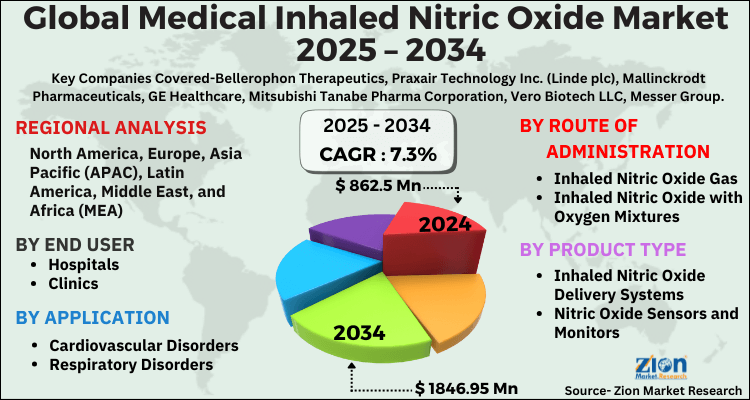
The global medical inhaled nitric oxide market size was worth around USD 862.5 Million in 2024 and is predicted to grow to around USD 1846.95 Million by 2034 with a compound annual growth rate (CAGR) of roughly 7.3% between 2025 and 2034. The report analyzes the global medical inhaled nitric oxide market's drivers, restraints/challenges, and the effect they have on the demands during the projection period. In addition, the report explores emerging opportunities in the medical inhaled nitric oxide industry.
Medical Inhaled Nitric Oxide Market: Overview
Medical inhaled nitric oxide is a specialized treatment within the pharmaceutical and medical sector, designed to address respiratory and cardiovascular issues. This therapeutic gas, composed of nitric oxide, is administered through inhalation to patients in controlled medical environments. It plays a crucial role in improving oxygenation and reducing pulmonary hypertension by selectively dilating pulmonary blood vessels. Medical inhaled nitric oxide is employed in various scenarios, including the treatment of pulmonary arterial hypertension (PAH), enhancing oxygenation in neonates with hypoxic respiratory failure, and assisting in managing adult respiratory distress syndrome (ARDS). Due to its precise administration requirements, this treatment is primarily utilized in hospitals, contributing significantly to advanced patient care and well-being.
Key Insights
- As per the analysis shared by our research analyst, the global medical inhaled nitric oxide market is estimated to grow annually at a CAGR of around 7.3% over the forecast period (2025-2034).
- Regarding revenue, the global medical inhaled nitric oxide market size was valued at around USD 862.5 Million in 2024 and is projected to reach USD 1846.95 Million by 2034.
- The medical inhaled nitric oxide market is projected to grow at a significant rate due to rising prevalence of respiratory diseases such as neonatal respiratory distress syndrome (RDS), acute respiratory distress syndrome (ARDS), and pulmonary hypertension, coupled with increasing awareness of its therapeutic benefits.
- Based on Product Type, the Inhaled Nitric Oxide Delivery Systems segment is expected to lead the global market.
- On the basis of Route of Administration, the Inhaled Nitric Oxide Gas segment is growing at a high rate and will continue to dominate the global market.
- Based on the Application, the Cardiovascular Disorders segment is projected to swipe the largest market share.
- By End User, the Hospitals segment is expected to dominate the global market.
- Based on region, North America is predicted to dominate the global market during the forecast period.
Medical Inhaled Nitric Oxide Market: Dynamics
Key Growth Drivers
The growth of the medical inhaled nitric oxide market is primarily driven by the rising prevalence of respiratory disorders such as neonatal hypoxemia, pulmonary hypertension, and chronic obstructive pulmonary disease (COPD). Increasing adoption of inhaled nitric oxide therapy in hospitals and intensive care units for managing oxygenation issues and improving pulmonary vasodilation is significantly boosting market expansion. Moreover, advancements in nitric oxide delivery systems, growing healthcare infrastructure, and increased awareness regarding the clinical benefits of inhaled nitric oxide are further propelling demand across both developed and emerging regions.
Restraints
Despite its promising outlook, the market faces several restraints, including the high cost of nitric oxide therapy and the associated delivery devices, which limit accessibility, particularly in low- and middle-income countries. Strict regulatory requirements and lengthy product approval processes also hinder market growth. Additionally, potential adverse effects of prolonged nitric oxide exposure, such as methemoglobinemia and nitrogen dioxide toxicity, pose safety concerns that can restrict its wider clinical adoption.
Opportunities
The market presents significant opportunities through technological innovations and the expanding application of inhaled nitric oxide in treating emerging conditions such as COVID-19-related respiratory distress and other acute lung injuries. Increasing research and development activities aimed at creating portable and cost-effective delivery systems are likely to enhance market penetration. Furthermore, growing investment in healthcare infrastructure and the expansion of homecare respiratory therapies in developing countries create new avenues for market growth in the coming years.
Challenges
The medical inhaled nitric oxide market faces challenges such as the need for continuous monitoring during therapy and the shortage of trained healthcare professionals capable of managing nitric oxide delivery systems. Competition from alternative treatment options for pulmonary conditions may also impact market growth. Additionally, maintaining gas stability and ensuring safe handling and storage conditions present technical challenges for manufacturers and healthcare providers, potentially affecting overall adoption rates.
Medical Inhaled Nitric Oxide Market: Segmentation
The global medical inhaled nitric oxide market is segmented based on product type, route of administration, end-user, and region. All the segments have been analyzed based on present and future trends and the market is estimated from 2025 to 2034.
Based on product type, the global market segments are inhaled nitric oxide delivery systems, nitric oxide sensors, and monitors. At present, the global market is dominated by the nitric oxide delivery systems segment. These systems encompass the equipment and mechanisms necessary for the controlled administration of inhaled nitric oxide to patients. They play a crucial role in regulating the concentration of nitric oxide delivered and ensuring its safe and effective use.
Based on route of administration, the global medical inhaled nitric oxide industry is categorized as inhaled nitric oxide gas, inhaled nitric oxide with oxygen mixtures. Out of these, inhaled nitric oxide gas was the largest shareholding segment in the global market. Inhaled nitric oxide gas typically leads in the medical inhaled nitric oxide market because it directly delivers pure nitric oxide for targeted therapeutic effects on pulmonary blood vessels, making it the primary mode of administration for desired outcomes.
Based on application, the global medical inhaled nitric oxide market is categorized as cardiovascular disorders and respiratory disorders. Out of these, the respiratory disorders segment was the largest shareholding segment in the global market. Inhaled nitric oxide gas generally leads in the application of respiratory disorders within the medical inhaled nitric oxide market, as its mechanism of improving oxygenation and reducing pulmonary hypertension aligns well with addressing respiratory challenges.
Based on end-user, the global medical inhaled nitric oxide industry is categorized as hospitals and clinics. Out of these, hospital segment was the largest shareholding segment in the global market due to the specialized resources and infrastructure required for administering this therapy effectively.
The Regional, this segment includes the current and forecast demand for North America, Europe, Asia Pacific, Latin America,and the Middle East and Africa.
Medical Inhaled Nitric Oxide Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Medical Inhaled Nitric Oxide Market |
| Market Size in 2024 | USD 862.5 Million |
| Market Forecast in 2034 | USD 1846.95 Million |
| Growth Rate | CAGR of 7.3% |
| Number of Pages | 214 |
| Key Companies Covered | Bellerophon Therapeutics, Praxair Technology Inc. (Linde plc), Mallinckrodt Pharmaceuticals, GE Healthcare, Mitsubishi Tanabe Pharma Corporation, Vero Biotech LLC, Messer Group, Air Liquide, Nu-Med Plus, Beyond Air Inc, and others. |
| Segments Covered | By Product Type, By Route of Administration, By Application, By End User, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, The Middle East and Africa (MEA) |
| Base Year | 2024 |
| Historical Year | 2020 to 2023 |
| Forecast Year | 2025 - 2034 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Medical Inhaled Nitric Oxide Market: Regional Analysis
North America, particularly the United States, stands out as a leading region in the global medical inhaled nitric oxide market. This prominence can be attributed to the region's advanced healthcare infrastructure, robust research & development capabilities, and a relatively high prevalence of conditions such as pulmonary arterial hypertension and respiratory distress syndrome that require specialized treatments like inhaled nitric oxide therapy. The United States, with its well-established healthcare system and extensive network of hospitals and medical centers, has been at the forefront of adopting cutting-edge medical technologies, contributing to its leadership position in the market.
Medical Inhaled Nitric Oxide Market: Competitive Analysis
The report provides a company market share analysis to give a broader overview of the key market players. In addition, the report also covers key strategic developments of the market, including acquisitions & mergers, new product launches, agreements, partnerships, collaborations & joint ventures, research & development, and regional expansion of major participants involved in the medical inhaled nitric oxide market on a global and regional basis.
The global medical inhaled nitric oxide market is dominated by players like:
- Bellerophon Therapeutics
- Praxair Technology, Inc. (Linde plc)
- Mallinckrodt Pharmaceuticals
- GE Healthcare
- Mitsubishi Tanabe Pharma Corporation
- Vero Biotech LLC
- Messer Group
- Air Liquide
- Nu-Med Plus
- Beyond Air, Inc.
The global medical inhaled nitric oxide market is segmented as follows:
By Product Type
- Inhaled Nitric Oxide Delivery Systems
- Nitric Oxide Sensors and Monitors
By Route of Administration
- Inhaled Nitric Oxide Gas
- Inhaled Nitric Oxide with Oxygen Mixtures
By Application
- Cardiovascular Disorders
- Respiratory Disorders
By End User
- Hospitals
- Clinics
By Region
- North America
- The U.S.
- Canada
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Table Of Content
Methodology
FrequentlyAsked Questions
Medical inhaled nitric oxide is a specialized treatment within the pharmaceutical and medical sector, designed to address respiratory and cardiovascular issues. This therapeutic gas, composed of nitric oxide, is administered through inhalation to patients in controlled medical environments.
The global medical inhaled nitric oxide market is expected to grow due to increasing neonatal ICU applications, rising prevalence of respiratory disorders, advancements in pulmonary hypertension treatments, and growing demand for critical care therapies.
According to a study, the global medical inhaled nitric oxide market size was worth around USD 862.5 Million in 2024 and is expected to reach USD 1846.95 Million by 2034.
The global medical inhaled nitric oxide market is expected to grow at a CAGR of 7.3% during the forecast period.
North America is expected to dominate the medical inhaled nitric oxide market over the forecast period.
Leading players in the global medical inhaled nitric oxide market include Bellerophon Therapeutics, Praxair Technology Inc. (Linde plc), Mallinckrodt Pharmaceuticals, GE Healthcare, Mitsubishi Tanabe Pharma Corporation, Vero Biotech LLC, Messer Group, Air Liquide, Nu-Med Plus, Beyond Air Inc, among others.
The report explores crucial aspects of the medical inhaled nitric oxide market, including a detailed discussion of existing growth factors and restraints, while also examining future growth opportunities and challenges that impact the market.
HappyClients