Global Chinese Hamster Ovary Cells (CHO) Market Size, Share, Growth Analysis Report - Forecast 2034

Chinese Hamster Ovary Cells (CHO) Market By Application (Clotting Factors, Monoclonal Antibodies, Hormones, Cytokines, Fc-Fusion Protein, Enzymes, Others), By Product (CHO-DG44, CHO-K1, CHO-S, CHO-DXB1, Others), By End-User (Contract Development & Manufacturing Organization, Biopharmaceutical Companies, Academic Institutes & Research, Biotech Companies, Clinical Research Organizations, Others), By System (Metabolic Selection System, Antibiotic Selection System), and By Region: Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, and Forecasts 2025 - 2034
| Market Size in 2024 | Market Forecast in 2034 | CAGR (in %) | Base Year |
|---|---|---|---|
| USD 273.15 Million | USD 484.12 Million | 5.89% | 2024 |
Chinese Hamster Ovary Cells (CHO) Market: Industry Perspective
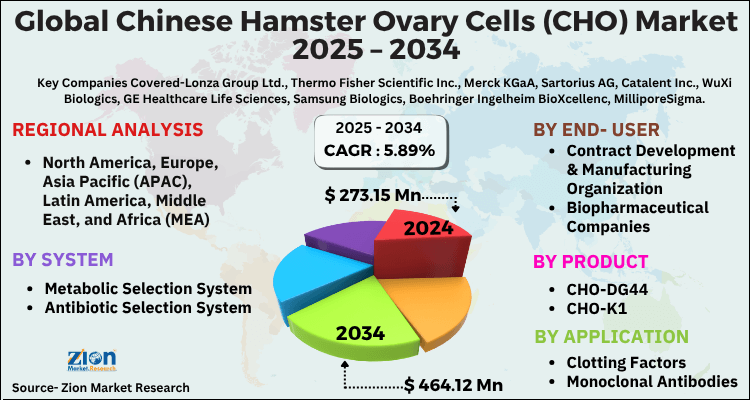
The global chinese hamster ovary cells (CHO) market size was worth around USD 273.15 Million in 2024 and is predicted to grow to around USD 484.12 Million by 2034 with a compound annual growth rate (CAGR) of roughly 5.89% between 2025 and 2034.
The report analyzes the global chinese hamster ovary cells (CHO) market's drivers, restraints/challenges, and the effect they have on the demands during the projection period. In addition, the report explores emerging opportunities in the chinese hamster ovary cells (CHO) industry.
The report delves deeper into several crucial aspects of the global Chinese hamster ovary cells (CHO) market. It includes a detailed discussion of existing growth factors and restraints. Future growth opportunities and challenges that impact the Chinese hamster ovary cells (CHO) industry are comprehensively addressed in the report.
Chinese Hamster Ovary Cells (CHO) Market: Overview
Chinese hamster ovary cells (CHO) are scientifically known as Cricetulus griseus and are cells derived from the ovaries of Chinese hamsters. They are used extensively in the pharmaceutical and biotechnology industries for the production of recombinant proteins and therapeutic proteins. CHO cells have been preferred for several research purposes since they have shown exceptional results in terms of growing well in culture. They also tend to produce large amounts of recombinant proteins and are known for relatively simpler growth requirements. Moreover, their stable genetic characteristics further add to the reasons working in the favor of demand and application of CHO cells. They are commonly used for the production of monoclonal antibodies, hormones, growth factors, enzymes, and other biopharmaceuticals. The CHO cell industry is likely to register steady growth in the coming years.
Key Insights
- As per the analysis shared by our research analyst, the global chinese hamster ovary cells (CHO) market is estimated to grow annually at a CAGR of around 5.89% over the forecast period (2025-2034).
- Regarding revenue, the global chinese hamster ovary cells (CHO) market size was valued at around USD 273.15 Million in 2024 and is projected to reach USD 484.12 Million by 2034.
- The chinese hamster ovary cells (CHO) market is projected to grow at a significant rate due to rising demand for biopharmaceuticals, advancements in recombinant protein production, increasing adoption in monoclonal antibody development, and growth in cell-based research and therapeutics.
- Based on Application, the Clotting Factors segment is expected to lead the global market.
- On the basis of Product, the CHO-DG44 segment is growing at a high rate and will continue to dominate the global market.
- Based on the End-User, the Contract Development & Manufacturing Organization segment is projected to swipe the largest market share.
- By System, the Metabolic Selection System segment is expected to dominate the global market.
- Based on region, North America is predicted to dominate the global market during the forecast period.
Chinese Hamster Ovary Cells (CHO) Market: Growth Drivers
Growing investment driving biopharmaceutical industry expansion to propel market growth
The global Chinese hamster ovary cells (CHO) market is projected to be driven by the increasing investment toward the expansion of the biopharmaceutical industry. In the last decade, the industry has become exceptionally important to the global healthcare and pharmaceutical sectors. Biopharmaceuticals have managed to provide answers to critical diseases which could not be cured using traditional medicines or therapies. The various components in the industry including biologic drugs, monoclonal antibodies, recombinant proteins, and gene therapies allow healthcare professionals to provide targeted therapies that are known to have a better success rate. Moreover, these therapies are being used to cure conditions such as cancer, autoimmune diseases, genetic disorders, and more. As the modern healthcare structure undertakes more research around biopharmaceuticals, the demand for CHO cells is likely to continue growing.
Chinese Hamster Ovary Cells (CHO) Market: Restraints
High cost of CHO cell-based production to restrict market expansion
Generally, the cost of CHO cell-based production processes tends to be higher than alternative expression systems such as microbial systems. CHO cells demand specialized growth media along with the use of an extremely complex purification process and extensive culture optimization. These factors are known to contribute to the overall cost of production which could limit the adoption of CHO cells for companies or research institutes working on a budget.
Chinese Hamster Ovary Cells (CHO) Market: Opportunities
Rising efforts toward higher production of biosimilars to provide growth opportunities
The Chinese hamster ovary cells industry is expected to come across several growth opportunities led by the growing interest and efforts of the medical community toward increasing the production of biosimilars. They are known to be cost-effective alternatives to reference biologics and a large section of biosimilars make use of CHO cells. With the increasing demand for quality medical care and the highest expense associated with it, governments and healthcare agencies are looking at measures to contain surging healthcare costs which could trigger more demand for biosimilars and in turn impact the consumption level of CHO cells.
Chinese Hamster Ovary Cells (CHO) Market: Challenges
Scaling up of CHO cell-based biopharmaceutical products to challenge market expansion
The CHO cell industry growth is expected to be challenged by the problems surrounding scaling up the production of CHO cell-based biopharmaceuticals. While working with larger scales of CHO cells, it may be difficult to achieve consistent and reproducible yields due to the requirement of process optimization, including optimizing culture conditions, media formulations, and bioreactor design. This may be a barrier for the companies operating in the CHO cells industry to ensure scalability while maintaining product quality and minimizing the cost of production.
Chinese Hamster Ovary Cells (CHO) Market: Segmentation
The global Chinese hamster ovary cells (CHO) market is segmented based on application, product, end-user, system, and region.
Based on application, the global market segments are clotting factors, monoclonal antibodies, hormones, cytokines, Fc-fusion protein, enzymes, and others. The industry registered the highest growth in the monoclonal antibodies (mAbs) segment in 2022 as CHO cells are used extensively for the production of mAbs. They are a class of biologic drugs that are used for the treatment of various diseases including infectious conditions, cancer, and autoimmune issues. Several beneficial attributes of CHO cells are the driving factors for a higher adoption rate. For instance, their ability to produce complex proteins with appropriate post-translational modifications, correct folding, and high yields. They offer advantages in terms of scalability, productivity, and regulatory acceptance. As of 2023, the global biopharmaceutical market is over USD 400 billion.
Based on product, the Chinese hamster ovary cells industry is segmented into CHO-DG44, CHO-K1, CHO-S, CHO-DXB1, and others.
Based on end-user, the global market divisions are contract development & manufacturing organization, biopharmaceutical companies, academic institute & research, biotech companies, clinical research organization, and others. The leading end-user of CHO cells in 2022 were known to be biopharmaceutical companies including larger corporations and small companies. They make use of CHO cells for the production of biologic drugs such as monoclonal antibodies, recombinant proteins, and other complex therapeutic molecules. In the industry, CHO cells have proven to offer a reliable and well-established platform for large-scale production of biopharmaceuticals. The academic institute & research centers may use CHO cells for studying protein expression, cell line engineering, process optimization, and other aspects of biopharmaceutical production. Between 2019 and 2022, the health research & development expenditure by the US federal government grew by USD 9.8 billion.
Based on the system, the global Chinese hamster ovary cells market is segmented into metabolic selection system and antibiotic selection system.
Recent Developments:
- In February 2023, the European Investment Bank (EIB) signed a deal with Icosagen AS, one of the leading biotechnology companies based in Estonia. The deal is worth €18 million and will be used for financing the production facility owned by Icosagen along with supporting the undertakings for strengthening the research & development technology platforms of the company
- In January 2021, Pfizer, a US-based multinational pharmaceutical giant and a biotechnology corporation, announced an investment of USD 120 million in biotechnology innovation. The announcement was made through the Pfizer Breakthrough Growth Initiative (PBGI), through which the company will be investing a total of up to USD 500 million in biotechnology companies along with access to its scientific expertise
Chinese Hamster Ovary Cells (CHO) Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Chinese Hamster Ovary Cells (CHO) Market |
| Market Size in 2024 | USD 273.15 Million |
| Market Forecast in 2034 | USD 484.12 Million |
| Growth Rate | CAGR of 5.89% |
| Number of Pages | 204 |
| Key Companies Covered | Lonza Group Ltd., Thermo Fisher Scientific Inc., Merck KGaA, Sartorius AG, Catalent Inc., WuXi Biologics, GE Healthcare Life Sciences, Samsung Biologics, Boehringer Ingelheim BioXcellenc, MilliporeSigma, Fujifilm Diosynth Biotechnologies, GenScript Biotech Corporation, AGC Biologics, Selexis SA, Rentschler Biopharma SE, Celonic AG, Abzena Ltd., ProBioGen AG, BioVectra Inc., Eurofins BioPharma Product Testing, Glycotope GmbH, Horizon Discovery Group plc, Invetech, Lonza Biologics, Fujifilm Irvine Scientific, and others. |
| Segments Covered | By Application, By Product, By End-User, By System, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, The Middle East and Africa (MEA) |
| Base Year | 2024 |
| Historical Year | 2020 to 2023 |
| Forecast Year | 2025 - 2034 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Chinese Hamster Ovary Cells (CHO) Market: Regional Analysis
North America to generate the highest revenue in the near future
The global Chinese hamster ovary cells (CHO) market is expected to register the highest growth in North America and particularly the United States. The steady growth trajectory is associated with the presence of a robust pharmaceutical and healthcare infrastructure in the US along with the presence of some of the biggest and most well-funded pharmaceutical companies that are constantly investing in revolutionizing the global biopharma segment. In addition to this, the US government has increased its total healthcare R&D expenditure along with an increased rate of investment toward regional healthcare infrastructure. The National Institute of Health spends the majority of its annual budget which currently stands at USD 45 billion on medical research. Additionally, the growing demand for targeted medical therapies and medications could lead to higher adoption of CHO cells for further research and medicine development.
Chinese Hamster Ovary Cells (CHO) Market: Competitive Analysis
The report provides a company market share analysis to give a broader overview of the key market players. In addition, the report also covers key strategic developments of the market, including acquisitions & mergers, new product launches, agreements, partnerships, collaborations & joint ventures, research & development, and regional expansion of major participants involved in the chinese hamster ovary cells (CHO) market on a global and regional basis.
The global chinese hamster ovary cells (CHO) market is dominated by players like:
- Lonza Group Ltd.
- Thermo Fisher Scientific Inc.
- Merck KGaA
- Sartorius AG
- Catalent Inc.
- WuXi Biologics
- GE Healthcare Life Sciences
- Samsung Biologics
- Boehringer Ingelheim BioXcellenc
- MilliporeSigma
- Fujifilm Diosynth Biotechnologies
- GenScript Biotech Corporation
- AGC Biologics
- Selexis SA
- Rentschler Biopharma SE
- Celonic AG
- Abzena Ltd.
- ProBioGen AG
- BioVectra Inc.
- Eurofins BioPharma Product Testing
- Glycotope GmbH
- Horizon Discovery Group plc
- Invetech
- Lonza Biologics
- Fujifilm Irvine Scientific
The global Chinese hamster ovary cells (CHO) market is segmented as follows:
By Application
- Clotting Factors
- Monoclonal Antibodies
- Hormones
- Cytokines
- Fc-Fusion Protein
- Enzymes
- Others
By Product
- CHO-DG44
- CHO-K1
- CHO-S
- CHO-DXB1
- Others
By End-User
- Contract Development & Manufacturing Organization
- Biopharmaceutical Companies
- Academic Institutes & Research
- Biotech Companies
- Clinical Research Organizations
- Others
By System
- Metabolic Selection System
- Antibiotic Selection System
By Region
- North America
- The U.S.
- Canada
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Table Of Content
Methodology
FrequentlyAsked Questions
The global chinese hamster ovary cells (CHO) market is expected to grow due to increasing demand for biologics and biosimilars, rising adoption in monoclonal antibody production, and growing advancements in cell line engineering and bioprocessing technologies.
According to a study, the global chinese hamster ovary cells (CHO) market size was worth around USD 273.15 Million in 2024 and is expected to reach USD 484.12 Million by 2034.
The global chinese hamster ovary cells (CHO) market is expected to grow at a CAGR of 5.89% during the forecast period.
North America is expected to dominate the chinese hamster ovary cells (CHO) market over the forecast period.
Leading players in the global chinese hamster ovary cells (CHO) market include Lonza Group Ltd., Thermo Fisher Scientific Inc., Merck KGaA, Sartorius AG, Catalent Inc., WuXi Biologics, GE Healthcare Life Sciences, Samsung Biologics, Boehringer Ingelheim BioXcellenc, MilliporeSigma, Fujifilm Diosynth Biotechnologies, GenScript Biotech Corporation, AGC Biologics, Selexis SA, Rentschler Biopharma SE, Celonic AG, Abzena Ltd., ProBioGen AG, BioVectra Inc., Eurofins BioPharma Product Testing, Glycotope GmbH, Horizon Discovery Group plc, Invetech, Lonza Biologics, Fujifilm Irvine Scientific, among others.
The report explores crucial aspects of the chinese hamster ovary cells (CHO) market, including a detailed discussion of existing growth factors and restraints, while also examining future growth opportunities and challenges that impact the market.
HappyClients

-market-size.png)