Global Remicade Biosimilar Market Size to Surpass USD 4.71 Billion at a CAGR of 5.4% Growth By 2034
07-Apr-2025 | Zion Market Research
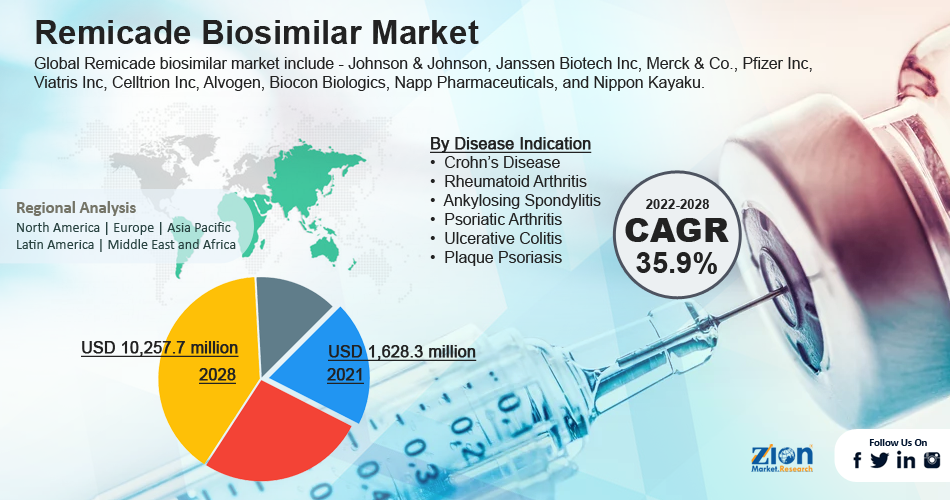
According to the report, the global remicade biosimilar market size was valued at approximately USD 2.82 Billion in 2024 and is predicted to reach around USD 4.71 Billion by the end of 2034, expanding at a CAGR of around 5.4% from 2025 to 2034.
The report analyzes the Remicade biosimilar market's drivers, restraints/challenges, and their effect on the demands during the projection period. In addition, the report explores emerging opportunities in the Remicade biosimilar market.
Biosimilars are biological products that are practically identical to an already FDA-approved drug, known as the reference biological product. Biosimilars are drugs approved by the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) and have no clinically significant differences from the source products regarding safety, purity, efficacy, and effectiveness.
One significant element enhancing the outlook for the Remicade biosimilar industry is the constant focus on developing biosimilar portfolios by internationally renowned pharmaceutical companies. In oncology, they are more likely to provide accessible natural therapeutic choices. There is a significant need for biological products to treat many ailments. According to the American Autoimmune Linked Disease Association, nearly 50 million Americans endured autoimmune diseases in 2018. The availability of these biosimilars in an injectable mode of administration, which results in faster reaction times, is estimated to grow the acceptance of Remicade biosimilars throughout the forecast period.
The global market for Remicade biosimilars is driven by efficient & advanced technology, enhanced patent expiration, increased public awareness of biosimilars, increased government initiatives worldwide, and the rising prevalence of chronic illnesses among the aging population. In addition, increased Remicade biosimilar adoption in emerging countries such as China, India, and others will develop new opportunities for the global Remicade biosimilars market. However, higher R&D costs and stringent government regulatory requirements are the primary restraints for market growth. Serious side effects of the drug that can require hospitalization or even be fatal are a barrier to the Remicade biosimilar.
These include invasive fungal infections, such as histoplasmosis, bacterial sepsis, tuberculosis, etc. Besides, the inherent complexity of the chemical compound and the absence of FDA-approved manufacturing facilities hinder the drug's market growth in developing regions.
The global Remicade biosimilar market is segregated based on disease indication and region. Based on disease indication, the market is divided into Crohn’s disease, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, ulcerative colitis, and plaque psoriasis. Among these, the rheumatoid arthritis segment led the market in 2021 and is expected to maintain its dominance throughout the forecast period.
The global Remicade biosimilar market is divided into geographic regions: North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Due to the existence of top market players in the region, the rapid entry of biosimilars into the market, and the high adoption rate owing to the biosimilars' low price, Europe is anticipated to have a dominating position in the worldwide market for Remicade biosimilar. For instance, Zessly (infliximab), a Remicade biosimilar, was approved for use in Europe in 2019 by the European Commission (EC), a division of Novartis.
This is primarily due to the bio-like version being introduced soon after the patent on the branded version had expired. Additionally, between 2022 and 2028, the market in the Asia Pacific is anticipated to develop at the greatest CAGR. A rapidly aging population, increased consumer awareness, favorable government policies, and rising prevalence of chronic illnesses in emerging nations such as China and India are driving the regional growth. Besides, significant investments by players in the region in biosimilar research and development are expected to drive market growth in the Asia Pacific.
Key players in the global Remicade biosimilar market include Johnson & Johnson, Janssen Biotech Inc, Merck & Co., Pfizer Inc, Viatris Inc, Celltrion Inc, Alvogen, Biocon Biologics, Napp Pharmaceuticals, and Nippon Kayaku.
Recent Development:
- May 2022: Abevmy, a bevacizumab drug, has been launched in Canada by Biocon Biologics and Viatris Inc. This drug was created by Roche and is biosimilar to Avastin. Health Canada has given it the go-ahead for 4 oncology indications. The launch of this drug assisted the company in further expanding its oncology portfolio in Canada, thus increasing patients' access to cost-effective biologic drugs for cancer treatment.
- July 2018, Merck & Co. announced the release of RENFLEXIS (infliximab), a Remicade biosimilar, in the United States for all indications.
Browse the full “Remicade Biosimilar Market By Disease Indication (Crohn’s disease, Rheumatoid Arthritis, Ankylosing Spondylitis, Psoriatic Arthritis, Ulcerative Colitis, Plaque Psoriasis) and By Region - Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, and Forecasts 2025 – 2034.” Report at https://www.zionmarketresearch.com/report/remicade-biosimilar-market
Global Remicade Biosimilar Market is segmented as follows:
By Disease Indication
- Crohn’s Disease
- Rheumatoid Arthritis
- Ankylosing Spondylitis
- Psoriatic Arthritis
- Ulcerative Colitis
- Plaque Psoriasis
By Region
- North America
- The U.S.
- Canada
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
About Us:
Zion Market Research is an obligated company. We create futuristic, cutting-edge, informative reports ranging from industry reports, the company reports to country reports. We provide our clients not only with market statistics unveiled by avowed private publishers and public organizations but also with vogue and newest industry reports along with pre-eminent and niche company profiles. Our database of market research reports comprises a wide variety of reports from cardinal industries. Our database is been updated constantly in order to fulfill our clients with prompt and direct online access to our database. Keeping in mind the client’s needs, we have included expert insights on global industries, products, and market trends in this database. Last but not the least, we make it our duty to ensure the success of clients connected to us—after all—if you do well, a little of the light shines on us.
Contact Us:
Zion Market Research
244 Fifth Avenue, Suite N202
New York, 10001, United States
Tel: +49-322 210 92714
USA/Canada Toll-Free No.1-855-465-4651
Email: sales@zionmarketresearch.com
Website: https://www.zionmarketresearch.com
Zion Market Research
Tel: +1 (302) 444-0166
USA/Canada Toll Free No.+1 (855) 465-4651
3rd Floor,
Mrunal Paradise, Opp Maharaja Hotel,
Pimple Gurav, Pune 411061,
Maharashtra, India
Phone No +91 7768 006 007, +91 7768 006 008
US OFFICE NO +1 (302) 444-0166
US/CAN TOLL FREE +1 (855) 465-4651
Email: sales@zionmarketresearch.com
We have secured system to process your transaction.
Our support available to help you 24 hours a day, five days a week.
Monday - Friday: 9AM - 6PM
Saturday - Sunday: Closed

