
Global Medical Exoskeleton Market Is Likely To Grow At A CAGR Value of Around 43.70 % By 2028
26-May-2022 | Zion Market Research
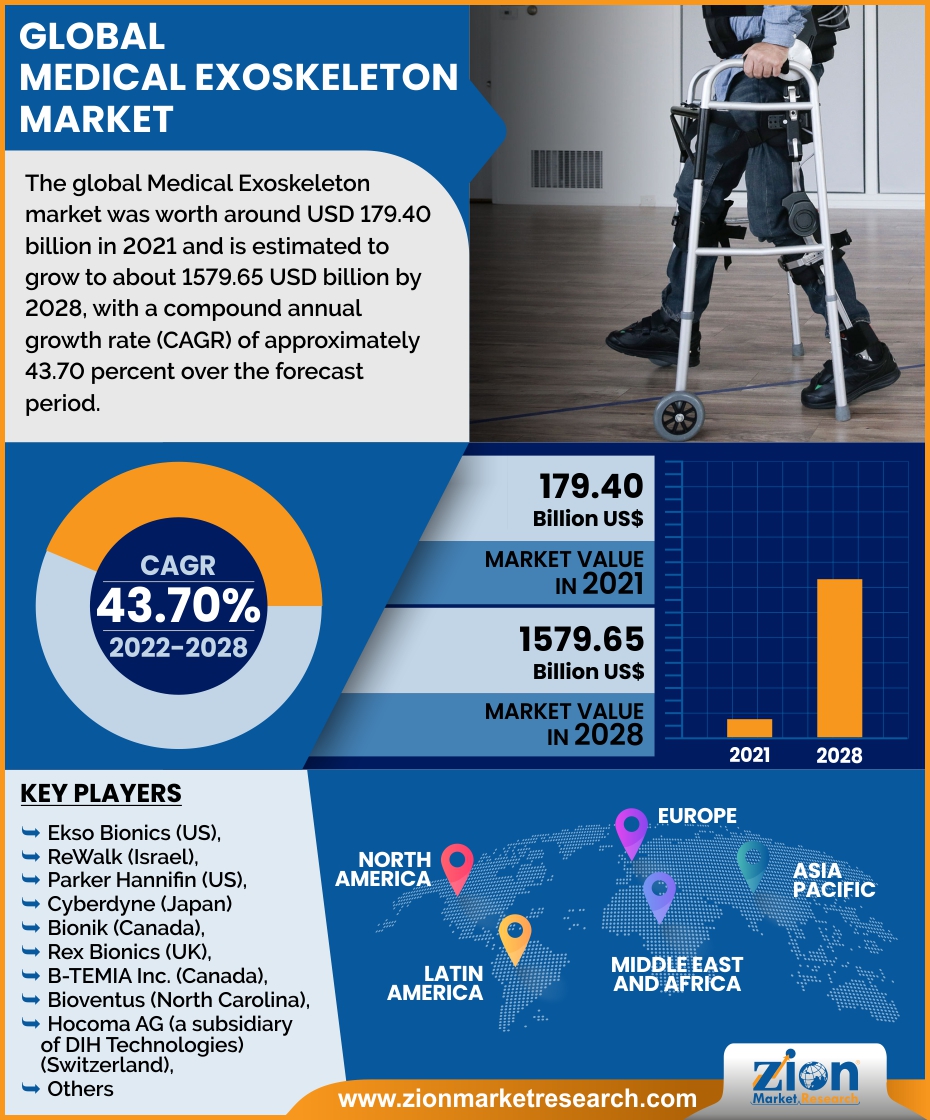
The global Medical Exoskeleton market was worth around USD 179.40 billion in 2021 and is estimated to grow to about USD 1579.65 billion by 2028, with a compound annual growth rate (CAGR) of approximately 43.70 percent over the forecast period. The report analyzes the digital remittance market’s drivers, restraints/challenges, and the effect they have on the demands during the projection period. In addition, the report explores emerging opportunities in the digital remittance market.
Exoskeletons are used to improve a person's physical strength by providing additional strength to limb movements. Medical exoskeletons assist physically disabled people, such as those who have suffered a spinal cord injury, suffer from a neurological ailment, are paralysed, or are elderly. Some medical human exoskeletons are used in physiotherapy and rehabilitation centres to treat the lower and upper extremities. The expansion of the global medical exoskeleton market is fueled by an increase in the number of orthopaedic operations, a growing geriatric population, and rising rates of road accidents. However, the product's high cost, as well as regulatory difficulties, is the main impediments to commercialization. As a result of this architecture, the limbs have greater mobility, strength, and endurance.
The demand for effective rehabilitation methods, which include the utilisation of cutting-edge technology and items, is growing around the world. This is viewed as a favourable indicator for the medical exoskeleton market's growth in all major regions. As a result, market dynamics may be affected during the predicted period. Furthermore, the growing number of people with physical disabilities will have a substantial impact on the market for medical exoskeletons. Furthermore, the growing senior population and increased access to medical exoskeleton insurance coverage in various countries are important drivers driving the medical exoskeleton market forward. Additionally, an increase in the number of traffic accidents, catastrophic injuries, and strokes will accelerate market expansion. One of the key industrial drivers that will force the medical exoskeleton market forward is increased spending on healthcare infrastructure.
Components, type, extremities, mobility, structure, and region are all used to segment the global Medical Exoskeleton market. The market is divided into hardware and software components. The hardware market is expected to have the greatest share by 2028. This is due to a growth in neurological, neurodegenerative, psychotic, and sleep disorders, as well as the requirement for early diagnosis, the availability of novel portable and wearable home-based monitoring devices, and increasing patient awareness. The market is classified into three categories: upper extremity medical exoskeletons, lower extremity medical exoskeletons, and complete body medical exoskeletons. In the estimated period, lower extremity medical exoskeletons are expected to account for a larger share of the market. Exoskeletons for the lower extremities give paraplegics and the elderly stability, weight-bearing, and movement capabilities. As a result, their utilisation of inpatient rehab is on the rise.
North America is expected to dominate the business in 2020, with a market share of 44.4 percent. The medical exoskeleton market in this region is being pushed by the expanding senior population, rising demand for self-assist exoskeletons, the high frequency of stroke, and the rising number of spinal cord injuries (SCI). Asia-Pacific is predicted to have a high CAGR in the global market due to an increase in the manufacturing of medical exoskeletons for the defense sector. Furthermore, China, Japan, and South Korea are expected to be the emerging world's top revenue generators..
Key players functioning in the global Medical Exoskeleton market include include Ekso Bionics (US), ReWalk (Israel), and Parker Hannifin (US), Cyberdyne (Japan) and Bionik (Canada), Rex Bionics (UK), B-TEMIA Inc. (Canada), Bioventus (North Carolina), Hocoma AG (a subsidiary of DIH Technologies) (Switzerland), Wearable Robotics SRL (Italy), Gogoa Mobility Robots SL (Spain), ExoAtlet (Luxembourg), Meditouch (Israel), Suit X (US), P&S Mechanics (South Korea), Marsi Bionics (Spain), Rehab Robotics (Hong Kong), Myomo (US), Focal Meditech (Netherlands), Honda Motors (Japan), Wandercraft (France), BAMA Teknoloji (Turkey), Medexo Robotics (Hong Kong), Fourier Intelligence (China), and TWIICE (Switzerland).
Recent developments:
- In March 2021, The Thai Food and Medical Products Approval Authority certified HAL Single Joint Type as a medical device (Thai FDA). This product is for patients who have reduced mobility in their upper and lower limbs owing to muscle weakness or paralysis..
Browse the full “Medical Exoskeleton Market by Component (Hardware and Software), By Type (Powered Exoskeletons and Passive Exoskeletons), By Extremity (Upper Extremity Medical Exoskeletons, Lower Extremity Medical Exoskeletons, Full Body), By Mobility (Mobile Exoskeletons and Stationary Exoskeletons), By Structure (Rigid Exoskeletons and Soft Exoskeleton) and By Region – Global and Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data and Forecasts 2022 – 2028.” Report at https://www.zionmarketresearch.com/report/medical-exoskeleton-market
Global Medical Exoskeleton market is segmented as follows:
By Component
- Hardware
- Software
By Type
- Powered Exoskeletons
- Passive Exoskeletons
By Extremity
- Upper Extremity Medical Exoskeletons
- Lower Extremity Medical Exoskeletons
- Full Body
By Mobility
- Mobile Exoskeletons
- Stationary Exoskeletons
By Structure
- Rigid Exoskeletons
- Soft Exoskeleton
By Region
- North America
- The U.S.
- Canada
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
About Us:
Zion Market Research is an obligated company. We create futuristic, cutting-edge, informative reports ranging from industry reports, the company reports to country reports. We provide our clients not only with market statistics unveiled by avowed private publishers and public organizations but also with vogue and newest industry reports along with pre-eminent and niche company profiles. Our database of market research reports comprises a wide variety of reports from cardinal industries. Our database is been updated constantly in order to fulfill our clients with prompt and direct online access to our database. Keeping in mind the client’s needs, we have included expert insights on global industries, products, and market trends in this database. Last but not the least, we make it our duty to ensure the success of clients connected to us—after all—if you do well, a little of the light shines on us.
Contact Us:
Zion Market Research
244 Fifth Avenue, Suite N202
New York, 10001, United States
Tel: +49-322 210 92714
USA/Canada Toll-Free No.1-855-465-4651
Email: sales@zionmarketresearch.com
Website: https://www.zionmarketresearch.com
