
Coronavirus Diagnostics Market To Register Exponential Growth By 2025
13-Mar-2020 | Zion Market Research
With the presence of a large pool of participants, the global coronavirus diagnostics market is displaying a highly competitive business landscape, finds a new research report by Zion Market Research (ZMR).
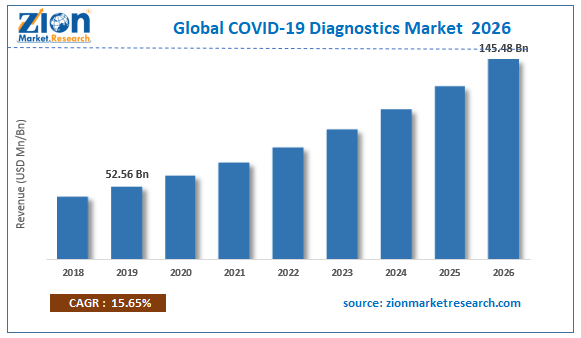
Browse the full “COVID-19 Diagnostics Market By Product (Instruments, Kits & Reagents, Diagnostic Services), By Technology (Molecular assay, Immunological assay, Digital PCR (dPCR), Clustered regularly interspaced short palindromic repeats (CRISPR), Microarray), By Test Type (Antigen-based Testing, Antibody (Serology) Testing, Molecular (PCR) Testing, Others) By Mode (Point-of-Care (PoC), Non-Point-of-Care (Non-PoC) By End-User (Hospitals, Diagnostic Labs & Clinics, Healthcare Labs & Clinics): Global Industry Perspective, Comprehensive Analysis and Forecast, 2020 - 2026.” Report at https://www.zionmarketresearch.com/report/coronavirus-diagnostics-market
Human coronaviruses are identified as key respiratory pathogens related to a myriad range of clinical ailments. Furthermore, these viruses are big, single-stranded, positive-sense RNA viruses that are the members of coronaviruses family. Moreover, two kinds of human coronaviruses, namely, CoV-229E and CoV-OC43- were discovered in the 1960s. However, the symptoms of these viruses were mild common cold and hence no particular attention was given to them.
Moreover, post-2000 - year, many new kinds of coronaviruses have emerged including SARS-CoV and MERS-CoV. Apart from this, in 2004 a novel coronavirus referred to as NL63 was detected in two children having respiratory problems across Holland and it was followed by the discovering of CoV-HKU1 in a patient suffering from pneumonia in the same country.
Research studies have demonstrated the presence of CoV-OC43 to be the most prominent coronavirus in countries prone to coronavirus. In addition to this, virus isolation in cell culture along with the molecular procedures particularly PCR or Polymerase Chain Reaction can help in identifying the CoV infections. An assay in the mariPOC respi test to identify species-particular CoV-OC43 nucleoprotein antigen was introduced in 2017. Reportedly, mariPOC is a multianalyte antigen detecting method that helps in quick identification of severe infections. Moreover, it can also diagnose nine respiratory disorders including influenza A, influenza B, adenovirus, parainfluenzavirus type 1 to 3, syncytial virus, human bocavirus, and human metapneumovirus. According to the study, the mariPOC respi test was found to have medical specificity of about 99.4% as compared to PCR tests in virus diagnosing.
According to NCBI, the assay assisted the researchers at NIH in successfully diagnosing the CoV-OC43 virus in six persons from Finland who were suffering from respiratory ailments. Furthermore, PCR tests on the samples of the patients conducted in two reputed labs in Holland also confirmed the same results.

With the onset of the recent novel coronavirus occurring in December 2019, the diagnosing, testing, and treatment of the disease is gaining significance across the globe. However, no particular antivirus therapy has been recommended by the WHO for 2019-nCoV. But, the global healthcare agency has provided certain guidelines for preventing the disease including non-invasive ventilation, protective mechanical ventilation, and oxygen therapy. Even the intake of lopinavir/ritonavir and alpha-interferon booster is allowed by WHO for the novel coronavirus patients.
For the record, in March 2020, Roche Diagnostics declared the U.S. FDA’s emergency use authorization of its cobas® systems for testing SARS-CoV-2. It is a qualitative detection of the new coronavirus referred to as SARS-CoV-2. As per the reports from authentic sources, hospitals and private labs across the globe can run completely automated cobas® 6800 & cobas® 8800 systems of Roche for diagnosis of the new coronavirus in patients.
Key players involved in the production of drugs for treating the coronavirus include Roche Diagnostics, Abbott, Bio-Rad Laboratories, Beckman Coulter, Becton, Dickinson and Company, Lonza, bioMerieux, Hologic, Qiagen, GSK Biologicals, PerkinElmer, and Ortho-Clinical Diagnostics.
By Region
- North America
- The U.S.
- Canada
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
About Us:
Zion Market Research is an obligated company. We create futuristic, cutting-edge, informative reports ranging from industry reports, the company reports to country reports. We provide our clients not only with market statistics unveiled by avowed private publishers and public organizations but also with vogue and newest industry reports along with pre-eminent and niche company profiles. Our database of market research reports comprises a wide variety of reports from cardinal industries. Our database is been updated constantly in order to fulfill our clients with prompt and direct online access to our database. Keeping in mind the client’s needs, we have included expert insights on global industries, products, and market trends in this database. Last but not the least, we make it our duty to ensure the success of clients connected to us—after all—if you do well, a little of the light shines on us.
Contact Us:
Zion Market Research
244 Fifth Avenue, Suite N202
New York, 10001, United States
Tel: +49-322 210 92714
USA/Canada Toll-Free No.1-855-465-4651
Email: sales@zionmarketresearch.com
Website: https://www.zionmarketresearch.com
