
Global Biologics Safety Testing Market Set For Robust Growth, To Reach Around USD 6.79 Billion by 2025
06-Nov-2019 | Zion Market Research
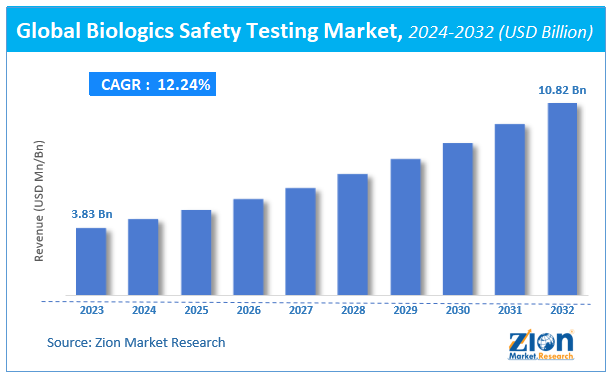
Zion Market Research has published a new report titled “Biologics Safety Testing Market by Product (Services, Instruments and Reagents and Kits); by Test Type (Bioburden tests, Adventitious Agent Detection Tests, Residual Host Contamination Detection Tests, Cell Line Authentication and Characterization Tests, Sterility Tests, Endotoxin Tests and Other Test Types); by Application (Tissue and Tissue-related Products Testing, Cellular and Gene Therapy, Blood and Blood-related Products Testing, Vaccine and Therapeutics Development and Stem Cell Research): Global Industry Perspective, Comprehensive Analysis and Forecast, 2018 – 2025”. According to the report, global demand for biologics safety testing market was valued at approximately USD 3.16 billion in 2018, and is expected to generate revenue of around USD 6.79 billion by end of 2025, growing at a CAGR of around 11.6% between 2019 and 2025.
Browse the full “Biologics Safety Testing Market: by Product (Services, Instruments and Reagents and Kits); by Test Type (Bioburden tests, Adventitious Agent Detection Tests, Residual Host Contamination Detection Tests, Cell Line Authentication and Characterization Tests, Sterility Tests, Endotoxin Tests and Other Test Types); by Application (Tissue and Tissue-related Products Testing, Cellular and Gene Therapy, Blood and Blood-related Products Testing, Vaccine and Therapeutics Development and Stem Cell Research): Global Industry Perspective, Comprehensive Analysis and Forecast, 2018 - 2025” Report at https://www.zionmarketresearch.com/report/biologics-safety-testing-market
Biologics drugs or products are derived from biological sources or are manufactured using living microorganisms, animal or plant cells. Majority of the biologics available in the market are derived using recombinant DNA technology. These biologics can be large or small polypeptides, products purified from serum and blood, antitoxins, vaccines, viruses, toxins, proteins, glycoproteins etc. Biologics have revolutionized the management and treatment of various chronic illnesses including cancer, rheumatoid arthritis, cardiovascular diseases, diabetes etc. Biologics safety is important concern and parameter for biologics manufacturer for regulatory compliance and use in animals and humans.
Global biologics safety testing market growth is supported by various factors such as increasing chronic disease burden and growing demand for biologics for treating diabetes, cancer, cardiovascular diseases and other chronic diseases, rapid growth in new product launches, increasing investments in life science R&D and growth in biopharmaceutical and pharmaceutical research. However, longer approval time and stringent regulatory requirements may act as restraining factors for the market growth. Emerging markets and growth in demand for pharmaceutical outsourcing are expected to bring new growth avenues for major market players in biologics safety testing.
The global biologics safety testing market is segmented based on product, technology, test type, application and region. Based on product, global biologics safety testing market is categorized into instruments, services and reagents and kits. Repeated and consistent use of reagents and kits led to largest market share of reagents and kits segment in 2018. Services segment is projected to register highest growth in the coming years as a result of growing demand for outsourcing services.
Based on test types biologics safety testing market is categorized into cell line authentication and characterization tests, adventitious agent detection, sterility, endotoxin, bioburden, residual host contamination detection tests, and other test types. The market was dominated by endotoxin test segment in 2018. Sterility test type segment will register highest CAGR in the coming years.
The application segment market is categorized into vaccine and therapeutics development, cellular and gene therapy, blood and blood-related products testing, stem cell research and tissue and tissue-related products testing. Growth in therapeutic drug and vaccine development projects by key pharmaceutical and biopharmaceutical companies has led to increasing adoption of biologics safety testing and thus accounted for largest market share in 2018.
North America held largest market share in 2018 for global biologics safety testing. Growing emphasis on novel therapeutics and vaccine development by biopharmaceutical and pharmaceutical companies, availability of funds, presence of developed R&D infrastructure, growing R&D investments, and technological advancements are factors driving the market in this region. Europe was second leading regional market. Increase in biologics development activities, high R&D spending, presence of developed infrastructure and government support for R&D are some of factors that boost market growth in this region. Latin America is estimated to register considerable growth in the coming years. Asia Pacific region will exhibit highest CAGR for biologics safety testing market in the coming years. Emphasis on capturing growth opportunities in developing nations by of contract research organizations, increasing chronic disease burden and increase in R&D spending are factors that promote biologics safety testing market growth in Asia Pacific. Lack of funds and infrastructure may act as restraining factors for market growth in Middle East and Africa.
Major players included in the report are Merck KGaA, SGS SA, Charles River Laboratories, Toxikon Corporation, Lonza Group LTD., Cytovance Biologics, Inc., Pace Analytical Services Inc., Sartorius AG, WuXi Apptec and Thermo Fisher Scientific Inc. among others.
The report segment of global biologics safety testing market as follows:
Global Biologics Safety Testing Market: By Product
- Reagents and Kits
- Services
- Instruments
Global Biologics Safety Testing Market: By Test Type
- Endotoxin Tests
- Sterility Tests
- Cell Line Authentication and Characterization Tests
- Residual Host Contamination Detection Tests
- Adventitious Agent Detection Tests
- Bioburden tests
- Other Tests
Global Biologics Safety Testing Market: By Application
- Vaccine and Therapeutics Development
- Blood and Blood-related Products Testing
- Cellular and Gene Therapy
- Tissue and Tissue-related Products Testing
- Stem Cell Research
Global Biologics Safety Testing Market: By Region
- North America
- U.S.
- Europe
- UK
- France
- Germany
- Asia Pacific
- China
- Japan
- India
- Latin America
- Brazil
- Middle East and Africa
About Us:
Zion Market Research is an obligated company. We create futuristic, cutting-edge, informative reports ranging from industry reports, the company reports to country reports. We provide our clients not only with market statistics unveiled by avowed private publishers and public organizations but also with vogue and newest industry reports along with pre-eminent and niche company profiles. Our database of market research reports comprises a wide variety of reports from cardinal industries. Our database is been updated constantly in order to fulfill our clients with prompt and direct online access to our database. Keeping in mind the client’s needs, we have included expert insights on global industries, products, and market trends in this database. Last but not the least, we make it our duty to ensure the success of clients connected to us—after all—if you do well, a little of the light shines on us.
Contact Us:
Zion Market Research
244 Fifth Avenue, Suite N202
New York, 10001, United States
Tel: +49-322 210 92714
USA/Canada Toll-Free No.1-855-465-4651
Email: sales@zionmarketresearch.com
Website: https://www.zionmarketresearch.com
